Abstract
Sporadic or hereditary colorectal cancer (CRC) with microsatellite instability (MSI) is frequently characterized by inflammatory lymphocytic infiltration and tends to be associated with a better outcome than microsatellite stable (MSS) CRC, probably reflecting a more effective immune response. We investigated inflammatory mechanisms in 48 MSI CRCs and 62 MSS CRCs by analyzing: (1) the expression of 48 cytokines using Bio-Plex multiplex cytokine assays, and (2) the in situ immune response by immunohistochemical analysis with antibodies against CD3 (T lymphocytes), CD8 (cytotoxic T lymphocytes), CD45RO (memory T lymphocytes), T-bet (Th1 CD4 cells), and FoxP3 (regulatory T cells). MSI CRC exhibited significantly higher expression of CCL5 (RANTES), CXCL8 (IL-8), CXCL9 (MIG), IL-1β, CXCL10 (IP-10), IL-16, CXCL1 (GROα), and IL-1ra, and lower expression of MIF, compared with MSS CRC. Immunohistochemistry combined with image analysis indicated that the density of CD3+, CD8+, CD45RO+, and T-bet+ T lymphocytes was higher in MSI CRC than in MSS CRC, whereas the number of regulatory T cells (FoxP3+) was not statistically different between the groups. These results indicate that MSI CRC is associated with a specific cytokine expression profile that includes CCL5, CXCL10, and CXCL9, which are involved in the T helper type 1 (Th1) response and in the recruitment of memory CD45RO+ T cells. Our findings highlight the major role of adaptive immunity in MSI CRC and provide a possible explanation for the more favorable prognosis of this CRC subtype.
Introduction
The immune contexture of solid tumors in humans has become an emerging hallmark of cancer and assessing its impact on clinical outcome might lead to the identification of new prognostic markers.Citation1,Citation2 Indeed, colorectal cancers (CRCs) that display a strong and coordinated adaptive immune response, as indicated by a high density of CD45RO+ memory and CD8+ cytotoxic T lymphocytes, are typically associated with a good prognosis.Citation3-Citation5
CRC is considered to be a heterogeneous disease. Approximately 85% of CRCs occur in a context of chromosomal instability and 15% display a deficiency in the DNA mismatch repair (MMR) systemCitation6,Citation7 linked to either epigenetic or genetic alterations. Defects in the DNA MMR machinery naturally give rise to microsatellite instability (MSI), a condition in which repetitive DNA sequences named microsatellites accumulate mutations that can affect tumor suppressor genes and oncogenes.Citation8 Although not specific to this disease, pronounced peritumoral lymphoid reaction (Crohn-like reaction) and dense infiltration of the tumor by lymphocytes are typically associated with MSI CRCCitation9,Citation10 and could contribute to its good prognosis.Citation7,Citation11 The immunogenicity of MSI CRC is attributed to the occurrence of microsatellite mutations that generate immunogenic neo-antigens.Citation12-Citation14
One mechanism involved in the recruitment of inflammatory cells at the lesion is the expression of cytokines, chemokines, and growth factors by cancer cells and the tumor microenvironment. In addition to its positive involvement in the identification and destruction of malignant cells, inflammation may also play an important role during cancer development and progression.Citation15-Citation17 Initiation of carcinogenesis,Citation18 tumor progression,Citation19 angiogenesis,Citation20,Citation21 and metastatic processesCitation22,Citation23 can be modulated by inflammation. Thus, inflammation appears to be a key process with dual functions mediating the relationship between cancer cells and the tumor microenvironment.Citation24 Recent studies have assessed the expression of various cytokines, chemokines, and their receptors in CRC, but these studies only focused on a limited number of factors and the tumor MMR status was rarely taken into consideration.Citation3,Citation25-Citation28 Therefore, in this study we aimed to delineate the differential role of inflammatory components and the tumor microenvironment in CRCs relative to their MMR status. To this end, we characterized the immune cell infiltrate in 62 microsatellite-stable (MSS) CRCs and 48 MSI CRCs by immunohistochemistry (IHC) and quantified their cytokine profile using multiplex-based assays.
Results
Clinicopathologic features
At the time of surgery, the median age was 72.5 y (range 30–95) for patients with MSI CRC and 65.0 y (range 30–86) for patients with MSS CRC (P = 0.206) (see ). The stage distribution of tumors was statistically different between groups (P = 0.018): 52% of MSI CRCs were classified as Stage II vs. 44% of MSS CRCs, whereas 29% of MSS CRCs were Stage IV vs. 6% of MSI CRCs. As expected, MSI CRC was more commonly identified in the right colon (65% were proximal to the splenic flexure, P = 0.018) and was more likely to be poorly differentiated than MSS CRC (35% vs. 8%, P = 0.004). MSI CRC also displayed a significantly higher level of lymphocytic infiltration (58% vs. 37%, P = 0.034) and an expansile tumor border configuration (55% vs. 33%, P = 0.040) as assessed by morphologic evaluation.
Table 1. Characteristics of the study population
Inflammatory infiltrates in MSS and MSI CRC
Immunohistochemical (IHC) analysis of tumor inflammatory cells showed a higher infiltration of CD3+ lymphocytes in the tumor center (ct) and invasive margin (im) areas of MSI CRC compared with MSS CRC (mean ± SD: 1335 ± 1320 vs. 777 ± 718 in the ct area, P = 0.046; 1574 ± 1017 vs. 1183 ± 1342 in the im area, P = 0.008). The number of CD8+ lymphocytes also was significantly increased in MSI CRC compared with MSS CRC (717 ± 724 vs. 262 ± 349 in the ct area, P < 0.001; 837 ± 629 vs. 539 ± 631 in the im area, P = 0.001), indicating efficient recruitment of cytotoxic cells ().
Figure 1. Distribution of immune cells in MSS and MSI colorectal cancers. (A–D) Comparison of the density of immune cells in human colorectal cancers with microsatellite stability (MSS; black bars) and microsatellite instability (MSI; gray bars). (A) The intratumoral (ct) and peritumoral (im) densities of CD3+, CD8+, T-Bet+, CD45RO+, FoxP3+, CD68+, and CD20+ cells were assessed by image analysis of tissue microarray spots. (B–D) Representative examples of staining are shown for CD8 (B), T-Bet (C), and FoxP3 (D); images show immunoperoxidase staining (×100) with the corresponding digital images (stained cells are represented in red). Statistical analyses were performed by non-parametric Mann-Whitney test; *P < 0.05; **P < 0.01; ***P < 0.001; ns, not significant
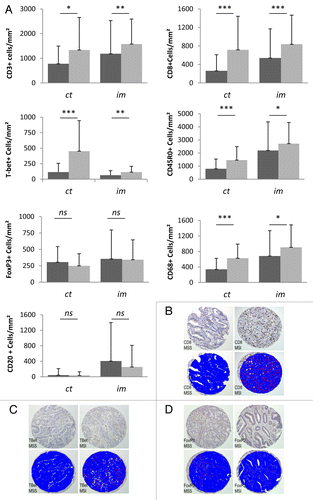
Because T helper type-1 (Th1) lymphocytes play a crucial role in activating cytotoxic T lymphocytes, we quantified the T-bet+ population, which is representative of the Th1 CD4+ T-cell subset. The density of T-bet+ cells in MSI CRC was significantly higher than that in MSS CRC for both the tumor center and invasive margin (453 ± 492 vs. 115 ± 141 in the ct area, P < 0.001; 115 ± 93 vs. 64 ± 74 in the im area, P = 0.001). Similarly, the mean number of CD45RO effector T cells was higher in MSI than in MSS CRC samples (1461 ± 1031 vs. 798 ± 743 in the ct area, P < 0.001; 2716 ± 1620 vs. 2195 ± 2186 in the im area, P = 0.025). On the other hand, FoxP3+ cells, which are representative of the regulatory T cell (Treg) population, were recruited to a similar extent in MSI and MSS CRC (250 ± 183 vs. 305 ± 237 in the ct area, P = 0.276; 343 ± 303 vs. 356 ± 441 in the im area, P = 0.490).
To determine whether other specific inflammatory populations were recruited, macrophages and B-lymphocytes were quantified using anti-CD68 and anti-CD20 antibodies, respectively. MSI CRC displayed a significantly higher number of CD68+ macrophages (626 ± 364 vs. 339 ± 285 in the ct area, P < 0.001; 908 ± 579 vs. 683 ± 653 in the im area, P = 0.019), whereas the density of tumor-infiltrating B cells was similar in both groups (36 ± 93 vs. 44 ± 168 in the ct area, P = 0.629; 255 ± 556 vs. 406 ± 993 in the im area, P = 0.712).
Cytokine expression in MSS and MSI CRC
We next measured cytokine expression using multiplex assays that allow the measurement of 48 cytokines. Many cytokines could not be detected (median = 0; IL-2, IL-4, IL-5, IL-9, IL-10, IL-13, IL-15, CCL3, G-CSF, TNFα, PDGFbb) or were barely detectable (median <1 pg/µg of total protein; IL-1α, IL2-Rα, IL-6, IL-7, IL-12 (p70), IL-17, IL-18, LIF, CCL11, CCL27, IFNγ, CCL2, CCL7, CCL4, β-FGF, β-NGF, IFN-α2, GM-CSF, M-CSF, SCF, TNFβ, TRAIL) in both MSS and MSI CRC protein samples ().
Table 2. Differential expression of cytokines, chemokines, and growth factors in MSS and MSS colorectal cancers
Among cytokines that were expressed, MSI CRC displayed a specific cytokine profile compared with MSS CRC: CCL5, CXCL8, CXCL9, IL-1β, CXCL10, IL-16, GROα, and IL-1ra were significantly overexpressed in MSI CRC, whereas the level of MIF was decreased (). CCL5, CXCL10, CXCL8, CXCL9, and IL-1β showed the strongest upregulation (between 12.9- and 2.3-fold) in MSI CRC compared with MSS CRC.
Finally, variations in cytokine expression within the MSS CRC group were analyzed by comparing MSS CRC with strong lymphocytic infiltration and/or Crohn-like lymphocytic reaction (inflammatory MSS CRC, n = 29) to MSS CRC without these features (non-inflammatory MSS CRC, n = 33). The initial classification into these two subgroups by morphologic evaluation was validated by IHC, which confirmed that the density of intra-tumoral CD3+ lymphocytes was higher in inflammatory than in non-inflammatory MSS CRC (882 ± 612 vs. 685 ± 798 cells/mm2, P = 0.044; ). Despite this significant difference, inflammatory and non-inflammatory MSS CRC had comparable cytokine contents. Conversely, although a similar density of CD3+ lymphocytes was observed in inflammatory MSS CRC and MSI CRC (882 ± 612 vs. 1335 ± 1320 CD3+ cells/mm2, P = 0.391), the levels of CCL5, CXCL8, CXCL9, IL-1β, CXCL10, IL-16, and IL-1ra remained significantly higher in the MSI group (). These data suggest that the distinct cytokine expression profile observed in MSI CRC is linked to the MSI status as well as the unique inflammatory infiltrate observed in this CRC subgroup.
Table 3. Inflammatory cell populations and cytokine expression in inflammatory MSS, non-inflammatory MSS, and MSI colorectal cancers
We also investigated the correlation between cytokine levels and specific immune cell densities in MSS and MSI groups. We identified significant correlations between different subsets of immune cells and chemokine expression, primarily in regards to CXCL9 and CXCL10 (Table S1). In MSS CRC, high levels of CXCL9 were associated with a significantly increased intratumoral density of CD3+, CD8+, and T-Bet+ T cells. Interestingly, the observed correlations between the frequency of infiltrating-immune cells and CXCL9 expression appeared stronger in MSI CRC in which such occurrences also seemed to involve the CD45RO population, and were not limited to the center of the tumor. Taken together, these data showed that in situ immune cells are strongly associated with a specific chemokine profile, indicating a distinct coordinated biological process integrating the immune system and the disease manifestations of CRC.
Discussion
The tumor microenvironment encompasses the morphologic and molecular features of constituent cancer cells, as well as those of non-malignant tumor cells, notably including immune cells that could, under key circumstances, play an important role in constraining CRC progression.Citation29,Citation30 Here, we show that MSI CRC displays a specific in situ immune response and chemokine profile in comparison to that of MSS CRC. This particular MSI-specific inflammatory microenvironment might explain the more favorable clinical course of this CRC subtype.
Using image analysis that allows objective quantification of the positive cells and minimizes the observer’s bias, we found a significant increase in CD3+, CD8+, CD45RO+, and T-bet+ lymphocytes in MSI CRC relative to MSS CRC, whereas the density of FoxP3+ cells was similar in both groups. These results are in agreement with previous studies that also reported a relationship between MSI and density of CD3+,Citation31-Citation33 CD8+,Citation26,Citation31,Citation33-Citation35 and CD45RO+ cells in CRC.Citation30,Citation35 Several studies have provided compelling evidence that effector/cytotoxic (CD3+ and CD8+) and memory (CD45RO+) T cells play major roles in the antitumor immune response in CRC, and, that their high expression correlates with a good clinical outcome (reviewed by Fridman et al.Citation2). CD8+ cytotoxic T lymphocytes can efficiently kill tumor cells and are mainly activated through the Th1 pathway. The Th1 pathway can be analyzed by assessing expression of the Tbox transcription factor T-bet, which is crucial for the development of effector Th1 CD4 T cellsCitation36 and is currently the most specific marker for this cell subset. T-bet mRNA levels and T-bet in situ protein expression in CRC have previously been correlated with reduced tumor recurrence.Citation5,Citation26 Here, we show for the first time that T-bet+ lymphocytes are significantly upregulated in MSI CRC, highlighting an efficient Th1 response that could account for the good clinical outcome of this population. Similarly, the increased density of CD45RO+ cells in MSI CRC in comparison to MSS CRC might enhance the efficiency of the antitumor immune response. Indeed, CD45RO+ cells, which include both antigen-exposed CD4+ lymphocytes and CD8+ lymphocytes, respond faster and with increased intensity to antigenic stimulation than do naive T cells.Citation2 Combined with the similar density of FoxP3+ cells found in MSI and MSS CRC, these results suggest that the lymphocyte balance is tipped in MSI CRC toward an effective host-mediated immune response rather than tolerance induction. Indeed, Tregs, which express the nuclear transcription factor FoxP3, dampen the antitumor immune response,Citation25,Citation33,Citation35,Citation37-Citation43 and suppress the activity of cytotoxic T cells (reviewed in deLeeuw et al.Citation44), thus maintaining immunologic tolerance. Few studies have evaluated the relationship between FoxP3+ and the MSI status, leading to controversial results in terms of prognosis.Citation25,Citation30,Citation33,Citation35,Citation40,Citation45,Citation46
It has been proposed that the local immune response in MSI CRC could be related to the production of potentially immunogenic neopeptides resulting from frameshift mutations within microsatellite sequences. Accordingly, Tougeron et al. have described a significant association between CD3+ density and the overall number of frameshift mutations.Citation47 The local inflammatory reaction evoked by cancer neoantigens preferentially arising in MSI CRC might thus promote cytokine production, which in turn, could expand the immune recruitment. On the other hand, the specific cytokine profile identified in this study involves mostly chemokines, specifically CXCL1, CXCL8, CXCL9, CXCL10, and CCL5. Based on the literature, these chemokines could, conceptually, be produced by immune cells. However, a number of colon cancer reports have shown these chemokines to be produced mainly by cancer cells and stromal cells, rather than by Th1, Th2, or Treg cells. Indeed, CXCL1 has been primarily detected in colon cancer cells and to a lesser extent in mesenchymal cells.Citation48,Citation49 Similar to CXCL10,Citation50,Citation51 CXCL8 has been shown to be primarily produced by carcinoma cellsCitation52,Citation53 but has also been detected at weaker levels in macrophages, lymphocytes, and myofibroblasts.Citation53 There are no publications describing the identity of the cells that produce CXCL9 in colon cancer, but the source could be neutrophilsCitation54 or M2 macrophages.Citation55 Finally, one report has suggested that CCL5 is produced by lymphocytes in colon cancer,Citation51 although CCL5 could be also produced by tumor-associated macrophages.Citation56 The correlations that we observed between various cytokines and specific subsets of immune cells at particular locations in the MSS or MSI tumors suggest a fine-tuned regulation of the in situ inflammatory recruitment, but whether the cytokine profile is a cause or consequence of immune infiltration remains unclear. However, chemokines within the tumor stroma are generally thought to play a role in the recruitment of immune cells. Some of these chemokines are characterized by antitumor activity, whereas others are either pro-tumorigenic or have a controversial role. Specifically, besides its chemoattractant properties for T lymphocytes, monocytes, natural killer cells, and eosinophils,Citation57 CCL5 has been shown to promote tumor growth and metastasis by inducing tumor cell proliferation, migration, angiogenesis, or expression of matrix metalloproteinases in various cancer types.Citation58-Citation61 Moreover, CCL5 can recruit Tregs within the tumor to kill cytotoxic CD8+ T cells,Citation62 suggesting that its overexpression could promote an immunosuppressive tumor microenvironment that might enhance tumor progression. Similarly, CXCL8 possesses tumorigenic and proangiogenic properties in CRC both in vitro and in vivo.Citation63 Nevertheless, in agreement with our study, Banerjea et al. described increased CXCL8 levels in MSI CRC.Citation64 Therefore, the overexpression of cytokines such as CCL5 and CXCL8 in MSI CRCs that are considered to have a relatively good prognosis suggests the presence of regulatory pathways that counterbalance their protumorigenic effects.
The Th1-type inflammatory mediators CXL9 and CXL10, two IFNγ-inducible CXCR3 ligands, act as angiostatic regulatorsCitation65 and promote the infiltration and expansion of antitumor T lymphocytes, particularly CD8+ effector T cellsCitation66,Citation67 and memory CD45RO+ T cells.Citation3 We found that tumors displaying high levels of CXCL9 and CXCL10 also showed significantly higher densities of CD3+, CD8+, and T-Bet+ cells, with stronger in situ recruitment in MSI tumors. Our results are in line with previous reports of a correlation between high CXCL9 and CXCL10 mRNA expression in CRC and increased density of CD8+, CD4+ cells and macrophages.Citation3,Citation50,Citation51 Moreover, these chemokines have been associated with better outcome.Citation3 Thus, together with the increased density of T-bet+ cells in MSI CRC, the higher expression levels of CXCL9 and CXCL10 suggest host protection via the generation of a Th1 immune response.
The role of CXCL1 in CRC progression is controversial. High CXCL1 expression has been associated with shorter recurrence-free survival in Stage III colorectal cancer patientsCitation27 and its downregulation results in a near-complete inhibition of tumor growth in nude mice.Citation68 However, CXCL1 transcription is higher in less invasive tumors and in samples from patients aged <65 y.Citation69 These results could be related to a stronger immune response in younger patients and to the fact that MSI CRCs are often early-stage tumors.
In summary, our data suggest a fine regulation of the immune contexture in MSI CRC, leading to efficient recruitment of inflammatory cells through the expression of specific chemokines. They also reveal a Th1-polarized immune response in MSI CRC through activation of the CXCL9/CXCL10 signaling axis. This translates into local recruitment or expansion of specific inflammatory populations that are involved in the anticancer response and potentiation of immunosurveillance, probably accounting for the favorable outcome of this particular subtype of colorectal cancer.
Patients and Methods
CRC samples and patients
CRC resection specimens with documented MMR status and available frozen tissue samples containing at least 50% tumor cells were identified at the pathology departments of the Val d’Aurelle Cancer Centre and Rouen University Hospital. In total, 110 CRC samples, of which 48 had MSI, were selected for this study. Among the 48 MSI CRCs, 11 were from patients with Lynch Syndrome, as defined by the presence of a deleterious germline mutation of a DNA damage repair gene involved in the MMR system. All samples were procured from fresh biopsies that were taken in the vicinity of the tumor invasion front and flash-frozen in liquid nitrogen. Tumor samples were collected following French laws under the supervision of an investigator and declared to the French Ministry of Higher Education and Research (declaration number DC-2008–695). All patients were informed about the use of their tissue samples for biological research and a written informed consent was systematically obtained for analysis of germline MMR gene mutations. The study was approved by the local translational research committee and was performed in accordance with the Helsinki Declaration of 1975. All samples were anonymized and analyses were performed blinded to the clinicopathologic data. Hematoxylin and eosin slides were reviewed by a gastrointestinal surgical pathologist (FB) to identify morphologic features, including histologic differentiation, lymphocytic infiltration, Crohn-like reaction, and tumor border configuration.Citation70 All tumors were staged according to the TMN classification system (7th edition) of the Union for International Cancer Control (UICC). The clinicopathologic features of the patients are reported in .
MMR status assessment
MMR status was assessed by IHC analysis for the expression of the hMLH1, hMSH2, hMSH6, and PMS2 proteins and by PCR analysis of microsatellites as previously described.Citation71
Protein extract preparation
Frozen CRC samples were sectioned into 15-μm slices to obtain 25–100 mg of tissue that was collected in Lysing Matrix D tubes (MP Biomedicals, # 116913500). Samples were crushed in TEG (10 mM TRIS-HCl, pH 7.4, 1.5 mM EDTA, and 10% glycerol) containing protease inhibitors (20 µg/mL aprotinin, 20 µg/mL leupeptin, 10 µg/mL pepstatin A, and 0.40 µg/mL phenylmethylsulfonyl fluoride) using a MagNA lyser (Roche Diagnostics) and then centrifuged at 13 000xg at 4 °C for 20 min. Total protein concentration in the supernatant was measured using the Bradford assay.
Bio-Plex multiplex cytokine assays
Two Bio-Plex ProTM Human kits (BioRad, #171-A11127 and #171-A11171) were used to measure the amount of cytokines, chemokines, and growth factors in CRC samples according to the manufacturer's instructions, as previously described.Citation72 The first multiplex assay detected 27 proteins (27-plex assay: IL-1β, IL-1ra, IL-2, IL-4, IL-5, IL-6, IL-7, CXCL8 [IL-8], IL-9, IL-10, IL-12 [p70], IL-13, IL-15, IL-17, CCL11 [Eotaxin], b-FGF, G-CSF, GM-CSF, IFNγ, CXCL10 [IP-10], CCL2 [MCP-1], CCL3 [MIP-1α], CCL4 [MIP-1β], PDGFbb, CCL5 [RANTES], TNFα, VEGF) and the second one detected 21 additional factors (21-plex assay: IL-1α, IL-2Ra, IL-3, IL-12 [p40], IL-16, IL-18, CCL27 [CTACK], CXCL1 [GRO-α], HGF, IFN-α2, LIF, CCL7 [MCP-3], M-CSF, MIF, CXCL9 [MIG], β-NGF, SCF, SCGF-β, CXCL12 [SDF-1α], TNFβ, TRAIL).
Coupled beads were incubated with 25 µg of total protein in a final volume of 50 µL. Data on the antibody reactions were acquired using the Bio-Plex system, which is a dual-laser, flow-based microplate reader system (BioRad). The concentrations of each target protein (expressed as pg/mL for 25 µg of total protein) were matched to the clinicopathologic data.
Tissue microarrays
After review of the archived tumor slides, tissue microarrays (TMAs) were prepared. Triplicate tissue cores (0.6 mm in diameter) were obtained from the tumor center (referred to as ct) and from the invasive margin (referred to as im), and arrayed using a manual arraying instrument (Beecher Instruments, MTA1).
Evaluation of tumor-infiltrating inflammatory cells
Tissue microarray sections were incubated with monoclonal antibodies against CD3 (clone LN10, Menarini), CD8 (clone C8/144B, Dako), CD45RO (clone UCHL1, Dako), FoxP3 (clone 236A/E7, AbCam), T-Bet (clone 4B10, SCB), CD20 (clone L26, Dako), and CD68 (clone KP1, Dako) on a Autostainer Link48 platform (Dako) using the Flex® system for signal amplification and diaminobenzidine tetrahydrochloride–chromogen (DAB) as a chromogen.
Immunoreactive cells were automatically quantified with the Spot Browser software (Excilone), as previously described.Citation5 Measurements were recorded as the number of positive cells per mm2 of tissue surface. Results were exported into an Excel file and data from triplicate cores were consolidated into a single score that was matched to the clinicopathologic data.
Statistical analysis
Continuous variables were described using mean, standard deviation, median, and range. For categorical variables, frequencies and percentages were computed. Possible associations between the microsatellite status and the clinicopathologic parameters were investigated using the χ2 test. The non-parametric Mann–Whitney test was used for continuous variables (quantification of cytokines and immunophenotypic markers). Differences were considered statistically significant when the P value was <0.05, except for the cytokine analyses for which the statistically significant threshold was corrected with the Bonferroni method to account for multiple testing and was set at 0.001. All statistical analyses were performed using STATA 10.0 (StataCorp).
| Abbreviations: | ||
| CRC | = | colorectal cancer |
| ct | = | tumor center |
| IHC | = | immunohistochemistry |
| im | = | invasive margin |
| MMR | = | mismatch repair |
| MSI | = | microsatellite instable |
| MSS | = | microsatellite stable |
| Th1 | = | T helper type-1 lymphocyte |
| TMA | = | tissue microarray |
| UICC | = | Union for International Cancer Control |
Additional material
Download Zip (138.9 KB)Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
Acknowledgments
The authors thank the tumor libraries “Tumorothèque” from ICM-Val d’Aurelle and Haute Normandie for the tumor samples. The study was funded by the Société Nationale Française de Gastroentérologie (SNFGE).
Citation: Boissière-Michot F, Lazennec G, Frugier H, Jarlier M, Roca L, Du Paty E, Laune D, Blanchard F, Le Pessot F, Sabourin JC, et al. Characterization of an adaptive immune response in microsatellite-instable colorectal cancer. OncoImmunology 2014; 3:e29256; 10.4161/onci.29256
References
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell 2011; 144:646 - 74; http://dx.doi.org/10.1016/j.cell.2011.02.013; PMID: 21376230
- Fridman WH, Pagès F, Sautès-Fridman C, Galon J. The immune contexture in human tumours: impact on clinical outcome. Nat Rev Cancer 2012; 12:298 - 306; http://dx.doi.org/10.1038/nrc3245; PMID: 22419253
- Mlecnik B, Tosolini M, Charoentong P, Kirilovsky A, Bindea G, Berger A, Camus M, Gillard M, Bruneval P, Fridman WH, et al. Biomolecular network reconstruction identifies T-cell homing factors associated with survival in colorectal cancer. Gastroenterology 2010; 138:1429 - 40; http://dx.doi.org/10.1053/j.gastro.2009.10.057; PMID: 19909745
- Pagès F, Berger A, Camus M, Sanchez-Cabo F, Costes A, Molidor R, Mlecnik B, Kirilovsky A, Nilsson M, Damotte D, et al. Effector memory T cells, early metastasis, and survival in colorectal cancer. N Engl J Med 2005; 353:2654 - 66; http://dx.doi.org/10.1056/NEJMoa051424; PMID: 16371631
- Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pagès C, Tosolini M, Camus M, Berger A, Wind P, et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 2006; 313:1960 - 4; http://dx.doi.org/10.1126/science.1129139; PMID: 17008531
- Jass JR. Classification of colorectal cancer based on correlation of clinical, morphological and molecular features. Histopathology 2007; 50:113 - 30; http://dx.doi.org/10.1111/j.1365-2559.2006.02549.x; PMID: 17204026
- Ogino S, Goel A. Molecular classification and correlates in colorectal cancer. J Mol Diagn 2008; 10:13 - 27; http://dx.doi.org/10.2353/jmoldx.2008.070082; PMID: 18165277
- Duval A, Rolland S, Compoint A, Tubacher E, Iacopetta B, Thomas G, Hamelin R. Evolution of instability at coding and non-coding repeat sequences in human MSI-H colorectal cancers. Hum Mol Genet 2001; 10:513 - 8; http://dx.doi.org/10.1093/hmg/10.5.513; PMID: 11181575
- Dolcetti R, Viel A, Doglioni C, Russo A, Guidoboni M, Capozzi E, Vecchiato N, Macrì E, Fornasarig M, Boiocchi M. High prevalence of activated intraepithelial cytotoxic T lymphocytes and increased neoplastic cell apoptosis in colorectal carcinomas with microsatellite instability. Am J Pathol 1999; 154:1805 - 13; http://dx.doi.org/10.1016/S0002-9440(10)65436-3; PMID: 10362805
- Phillips SM, Banerjea A, Feakins R, Li SR, Bustin SA, Dorudi S. Tumour-infiltrating lymphocytes in colorectal cancer with microsatellite instability are activated and cytotoxic. Br J Surg 2004; 91:469 - 75; http://dx.doi.org/10.1002/bjs.4472; PMID: 15048750
- Sargent DJ, Marsoni S, Monges G, Thibodeau SN, Labianca R, Hamilton SR, French AJ, Kabat B, Foster NR, Torri V, et al. Defective mismatch repair as a predictive marker for lack of efficacy of fluorouracil-based adjuvant therapy in colon cancer. J Clin Oncol 2010; 28:3219 - 26; http://dx.doi.org/10.1200/JCO.2009.27.1825; PMID: 20498393
- Linnebacher M, Gebert J, Rudy W, Woerner S, Yuan YP, Bork P, von Knebel Doeberitz M. Frameshift peptide-derived T-cell epitopes: a source of novel tumor-specific antigens. Int J Cancer 2001; 93:6 - 11; http://dx.doi.org/10.1002/ijc.1298; PMID: 11391614
- Schwitalle Y, Kloor M, Eiermann S, Linnebacher M, Kienle P, Knaebel HP, Tariverdian M, Benner A, von Knebel Doeberitz M. Immune response against frameshift-induced neopeptides in HNPCC patients and healthy HNPCC mutation carriers. Gastroenterology 2008; 134:988 - 97; http://dx.doi.org/10.1053/j.gastro.2008.01.015; PMID: 18395080
- Saeterdal I, Bjørheim J, Lislerud K, Gjertsen MK, Bukholm IK, Olsen OC, Nesland JM, Eriksen JA, Møller M, Lindblom A, et al. Frameshift-mutation-derived peptides as tumor-specific antigens in inherited and spontaneous colorectal cancer. Proc Natl Acad Sci U S A 2001; 98:13255 - 60; http://dx.doi.org/10.1073/pnas.231326898; PMID: 11687624
- Lazennec G, Richmond A. Chemokines and chemokine receptors: new insights into cancer-related inflammation. Trends Mol Med 2010; 16:133 - 44; http://dx.doi.org/10.1016/j.molmed.2010.01.003; PMID: 20163989
- Ali S, Lazennec G. Chemokines: novel targets for breast cancer metastasis. Cancer Metastasis Rev 2007; 26:401 - 20; http://dx.doi.org/10.1007/s10555-007-9073-z; PMID: 17717637
- Vindrieux D, Escobar P, Lazennec G. Emerging roles of chemokines in prostate cancer. Endocr Relat Cancer 2009; 16:663 - 73; http://dx.doi.org/10.1677/ERC-09-0109; PMID: 19556286
- Colotta F, Allavena P, Sica A, Garlanda C, Mantovani A. Cancer-related inflammation, the seventh hallmark of cancer: links to genetic instability. Carcinogenesis 2009; 30:1073 - 81; http://dx.doi.org/10.1093/carcin/bgp127; PMID: 19468060
- Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature 2008; 454:436 - 44; http://dx.doi.org/10.1038/nature07205; PMID: 18650914
- Strieter RM, Burdick MD, Mestas J, Gomperts B, Keane MP, Belperio JA. Cancer CXC chemokine networks and tumour angiogenesis. Eur J Cancer 2006; 42:768 - 78; http://dx.doi.org/10.1016/j.ejca.2006.01.006; PMID: 16510280
- Mehrad B, Keane MP, Strieter RM. Chemokines as mediators of angiogenesis. Thromb Haemost 2007; 97:755 - 62; PMID: 17479186
- Coussens LM, Werb Z. Inflammation and cancer. Nature 2002; 420:860 - 7; http://dx.doi.org/10.1038/nature01322; PMID: 12490959
- Zlotnik A, Burkhardt AM, Homey B. Homeostatic chemokine receptors and organ-specific metastasis. Nat Rev Immunol 2011; 11:597 - 606; http://dx.doi.org/10.1038/nri3049; PMID: 21866172
- Allen M, Louise Jones J. Jekyll and Hyde: the role of the microenvironment on the progression of cancer. J Pathol 2011; 223:162 - 76; http://dx.doi.org/10.1002/path.2803; PMID: 21125673
- Le Gouvello S, Bastuji-Garin S, Aloulou N, Mansour H, Chaumette MT, Berrehar F, Seikour A, Charachon A, Karoui M, Leroy K, et al. High prevalence of Foxp3 and IL17 in MMR-proficient colorectal carcinomas. Gut 2008; 57:772 - 9; http://dx.doi.org/10.1136/gut.2007.123794; PMID: 17965063
- Tosolini M, Kirilovsky A, Mlecnik B, Fredriksen T, Mauger S, Bindea G, Berger A, Bruneval P, Fridman WH, Pagès F, et al. Clinical impact of different classes of infiltrating T cytotoxic and helper cells (Th1, th2, treg, th17) in patients with colorectal cancer. Cancer Res 2011; 71:1263 - 71; http://dx.doi.org/10.1158/0008-5472.CAN-10-2907; PMID: 21303976
- Oladipo O, Conlon S, O’Grady A, Purcell C, Wilson C, Maxwell PJ, Johnston PG, Stevenson M, Kay EW, Wilson RH, et al. The expression and prognostic impact of CXC-chemokines in stage II and III colorectal cancer epithelial and stromal tissue. Br J Cancer 2011; 104:480 - 7; http://dx.doi.org/10.1038/sj.bjc.6606055; PMID: 21285972
- Halama N, Braun M, Kahlert C, Spille A, Quack C, Rahbari N, Koch M, Weitz J, Kloor M, Zoernig I, et al. Natural killer cells are scarce in colorectal carcinoma tissue despite high levels of chemokines and cytokines. Clin Cancer Res 2011; 17:678 - 89; http://dx.doi.org/10.1158/1078-0432.CCR-10-2173; PMID: 21325295
- Pagès F, Galon J, Fridman WH. The essential role of the in situ immune reaction in human colorectal cancer. J Leukoc Biol 2008; 84:981 - 7; http://dx.doi.org/10.1189/jlb.1107773; PMID: 18559950
- Nosho K, Baba Y, Tanaka N, Shima K, Hayashi M, Meyerhardt JA, Giovannucci E, Dranoff G, Fuchs CS, Ogino S. Tumour-infiltrating T-cell subsets, molecular changes in colorectal cancer, and prognosis: cohort study and literature review. J Pathol 2010; 222:350 - 66; http://dx.doi.org/10.1002/path.2774; PMID: 20927778
- Guidoboni M, Gafà R, Viel A, Doglioni C, Russo A, Santini A, Del Tin L, Macrì E, Lanza G, Boiocchi M, et al. Microsatellite instability and high content of activated cytotoxic lymphocytes identify colon cancer patients with a favorable prognosis. Am J Pathol 2001; 159:297 - 304; http://dx.doi.org/10.1016/S0002-9440(10)61695-1; PMID: 11438476
- Laghi L, Bianchi P, Miranda E, Balladore E, Pacetti V, Grizzi F, Allavena P, Torri V, Repici A, Santoro A, et al. CD3+ cells at the invasive margin of deeply invading (pT3-T4) colorectal cancer and risk of post-surgical metastasis: a longitudinal study. Lancet Oncol 2009; 10:877 - 84; http://dx.doi.org/10.1016/S1470-2045(09)70186-X; PMID: 19656725
- Michel S, Benner A, Tariverdian M, Wentzensen N, Hoefler P, Pommerencke T, Grabe N, von Knebel Doeberitz M, Kloor M. High density of FOXP3-positive T cells infiltrating colorectal cancers with microsatellite instability. Br J Cancer 2008; 99:1867 - 73; http://dx.doi.org/10.1038/sj.bjc.6604756; PMID: 18985040
- Prall F, Dührkop T, Weirich V, Ostwald C, Lenz P, Nizze H, Barten M. Prognostic role of CD8+ tumor-infiltrating lymphocytes in stage III colorectal cancer with and without microsatellite instability. Hum Pathol 2004; 35:808 - 16; http://dx.doi.org/10.1016/j.humpath.2004.01.022; PMID: 15257543
- Salama P, Phillips M, Grieu F, Morris M, Zeps N, Joseph D, Platell C, Iacopetta B. Tumor-infiltrating FOXP3+ T regulatory cells show strong prognostic significance in colorectal cancer. J Clin Oncol 2009; 27:186 - 92; http://dx.doi.org/10.1200/JCO.2008.18.7229; PMID: 19064967
- Mullen AC, High FA, Hutchins AS, Lee HW, Villarino AV, Livingston DM, Kung AL, Cereb N, Yao TP, Yang SY, et al. Role of T-bet in commitment of TH1 cells before IL-12-dependent selection. Science 2001; 292:1907 - 10; http://dx.doi.org/10.1126/science.1059835; PMID: 11397944
- Frey DM, Droeser RA, Viehl CT, Zlobec I, Lugli A, Zingg U, Oertli D, Kettelhack C, Terracciano L, Tornillo L. High frequency of tumor-infiltrating FOXP3(+) regulatory T cells predicts improved survival in mismatch repair-proficient colorectal cancer patients. Int J Cancer 2010; 126:2635 - 43; PMID: 19856313
- Pagès F, Kirilovsky A, Mlecnik B, Asslaber M, Tosolini M, Bindea G, Lagorce C, Wind P, Marliot F, Bruneval P, et al. In situ cytotoxic and memory T cells predict outcome in patients with early-stage colorectal cancer. J Clin Oncol 2009; 27:5944 - 51; http://dx.doi.org/10.1200/JCO.2008.19.6147; PMID: 19858404
- Suzuki H, Chikazawa N, Tasaka T, Wada J, Yamasaki A, Kitaura Y, Sozaki M, Tanaka M, Onishi H, Morisaki T, et al. Intratumoral CD8(+) T/FOXP3 (+) cell ratio is a predictive marker for survival in patients with colorectal cancer. Cancer Immunol Immunother 2010; 59:653 - 61; http://dx.doi.org/10.1007/s00262-009-0781-9; PMID: 19908042
- Correale P, Rotundo MS, Del Vecchio MT, Remondo C, Migali C, Ginanneschi C, Tsang KY, Licchetta A, Mannucci S, Loiacono L, et al. Regulatory (FoxP3+) T-cell tumor infiltration is a favorable prognostic factor in advanced colon cancer patients undergoing chemo or chemoimmunotherapy. J Immunother 2010; 33:435 - 41; http://dx.doi.org/10.1097/CJI.0b013e3181d32f01; PMID: 20386463
- Curiel TJ. Tregs and rethinking cancer immunotherapy. J Clin Invest 2007; 117:1167 - 74; http://dx.doi.org/10.1172/JCI31202; PMID: 17476346
- Chaput N, Louafi S, Bardier A, Charlotte F, Vaillant JC, Ménégaux F, Rosenzwajg M, Lemoine F, Klatzmann D, Taieb J. Identification of CD8+CD25+Foxp3+ suppressive T cells in colorectal cancer tissue. Gut 2009; 58:520 - 9; http://dx.doi.org/10.1136/gut.2008.158824; PMID: 19022917
- Blatner NR, Bonertz A, Beckhove P, Cheon EC, Krantz SB, Strouch M, Weitz J, Koch M, Halverson AL, Bentrem DJ, et al. In colorectal cancer mast cells contribute to systemic regulatory T-cell dysfunction. Proc Natl Acad Sci U S A 2010; 107:6430 - 5; http://dx.doi.org/10.1073/pnas.0913683107; PMID: 20308560
- deLeeuw RJ, Kost SE, Kakal JA, Nelson BH. The prognostic value of FoxP3+ tumor-infiltrating lymphocytes in cancer: a critical review of the literature. Clin Cancer Res 2012; 18:3022 - 9; http://dx.doi.org/10.1158/1078-0432.CCR-11-3216; PMID: 22510350
- Sinicrope FA, Rego RL, Ansell SM, Knutson KL, Foster NR, Sargent DJ. Intraepithelial effector (CD3+)/regulatory (FoxP3+) T-cell ratio predicts a clinical outcome of human colon carcinoma. Gastroenterology 2009; 137:1270 - 9; http://dx.doi.org/10.1053/j.gastro.2009.06.053; PMID: 19577568
- Lee WS, Park S, Lee WY, Yun SH, Chun HK. Clinical impact of tumor-infiltrating lymphocytes for survival in stage II colon cancer. Cancer 2010; 116:5188 - 99; http://dx.doi.org/10.1002/cncr.25293; PMID: 20665489
- Tougeron D, Fauquembergue E, Rouquette A, Le Pessot F, Sesboüé R, Laurent M, Berthet P, Mauillon J, Di Fiore F, Sabourin JC, et al. Tumor-infiltrating lymphocytes in colorectal cancers with microsatellite instability are correlated with the number and spectrum of frameshift mutations. Mod Pathol 2009; 22:1186 - 95; http://dx.doi.org/10.1038/modpathol.2009.80; PMID: 19503063
- Rubie C, Frick VO, Wagner M, Schuld J, Gräber S, Brittner B, Bohle RM, Schilling MK. ELR+ CXC chemokine expression in benign and malignant colorectal conditions. BMC Cancer 2008; 8:178; http://dx.doi.org/10.1186/1471-2407-8-178; PMID: 18578857
- Wang D, Wang H, Brown J, Daikoku T, Ning W, Shi Q, Richmond A, Strieter R, Dey SK, DuBois RN. CXCL1 induced by prostaglandin E2 promotes angiogenesis in colorectal cancer. J Exp Med 2006; 203:941 - 51; http://dx.doi.org/10.1084/jem.20052124; PMID: 16567391
- Jiang Z, Xu Y, Cai S. CXCL10 expression and prognostic significance in stage II and III colorectal cancer. Mol Biol Rep 2010; 37:3029 - 36; http://dx.doi.org/10.1007/s11033-009-9873-z; PMID: 19821051
- Musha H, Ohtani H, Mizoi T, Kinouchi M, Nakayama T, Shiiba K, Miyagawa K, Nagura H, Yoshie O, Sasaki I. Selective infiltration of CCR5(+)CXCR3(+) T lymphocytes in human colorectal carcinoma. Int J Cancer 2005; 116:949 - 56; http://dx.doi.org/10.1002/ijc.21135; PMID: 15856455
- Brew R, Southern SA, Flanagan BF, McDicken IW, Christmas SE. Detection of interleukin-8 mRNA and protein in human colorectal carcinoma cells. Eur J Cancer 1996; 32A:2142 - 7; http://dx.doi.org/10.1016/S0959-8049(96)00221-3; PMID: 9014758
- Cui G, Yuan A, Goll R, Vonen B, Florholmen J. Dynamic changes of interleukin-8 network along the colorectal adenoma-carcinoma sequence. Cancer Immunol Immunother 2009; 58:1897 - 905; http://dx.doi.org/10.1007/s00262-009-0702-y; PMID: 19350238
- Mantovani A, Cassatella MA, Costantini C, Jaillon S. Neutrophils in the activation and regulation of innate and adaptive immunity. Nat Rev Immunol 2011; 11:519 - 31; http://dx.doi.org/10.1038/nri3024; PMID: 21785456
- Galdiero MR, Garlanda C, Jaillon S, Marone G, Mantovani A. Tumor associated macrophages and neutrophils in tumor progression. J Cell Physiol 2013; 228:1404 - 12; http://dx.doi.org/10.1002/jcp.24260; PMID: 23065796
- Balkwill F. Cancer and the chemokine network. Nat Rev Cancer 2004; 4:540 - 50; http://dx.doi.org/10.1038/nrc1388; PMID: 15229479
- Homey B, Müller A, Zlotnik A. Chemokines: agents for the immunotherapy of cancer?. Nat Rev Immunol 2002; 2:175 - 84; http://dx.doi.org/10.1038/nri748; PMID: 11913068
- Mrowietz U, Schwenk U, Maune S, Bartels J, Küpper M, Fichtner I, Schröder JM, Schadendorf D. The chemokine RANTES is secreted by human melanoma cells and is associated with enhanced tumour formation in nude mice. Br J Cancer 1999; 79:1025 - 31; http://dx.doi.org/10.1038/sj.bjc.6690164; PMID: 10098731
- Sugasawa H, Ichikura T, Kinoshita M, Ono S, Majima T, Tsujimoto H, Chochi K, Hiroi S, Takayama E, Saitoh D, et al. Gastric cancer cells exploit CD4+ cell-derived CCL5 for their growth and prevention of CD8+ cell-involved tumor elimination. Int J Cancer 2008; 122:2535 - 41; http://dx.doi.org/10.1002/ijc.23401; PMID: 18246596
- Soria G, Ben-Baruch A. The inflammatory chemokines CCL2 and CCL5 in breast cancer. Cancer Lett 2008; 267:271 - 85; http://dx.doi.org/10.1016/j.canlet.2008.03.018; PMID: 18439751
- Cambien B, Richard-Fiardo P, Karimdjee BF, Martini V, Ferrua B, Pitard B, Schmid-Antomarchi H, Schmid-Alliana A. CCL5 neutralization restricts cancer growth and potentiates the targeting of PDGFRβ in colorectal carcinoma. PLoS One 2011; 6:e28842; http://dx.doi.org/10.1371/journal.pone.0028842; PMID: 22205974
- Chang LY, Lin YC, Mahalingam J, Huang CT, Chen TW, Kang CW, Peng HM, Chu YY, Chiang JM, Dutta A, et al. Tumor-derived chemokine CCL5 enhances TGF-β-mediated killing of CD8(+) T cells in colon cancer by T-regulatory cells. Cancer Res 2012; 72:1092 - 102; http://dx.doi.org/10.1158/0008-5472.CAN-11-2493; PMID: 22282655
- Ning Y, Manegold PC, Hong YK, Zhang W, Pohl A, Lurje G, Winder T, Yang D, LaBonte MJ, Wilson PM, et al. Interleukin-8 is associated with proliferation, migration, angiogenesis and chemosensitivity in vitro and in vivo in colon cancer cell line models. Int J Cancer 2011; 128:2038 - 49; http://dx.doi.org/10.1002/ijc.25562; PMID: 20648559
- Banerjea A, Ahmed S, Hands RE, Huang F, Han X, Shaw PM, Feakins R, Bustin SA, Dorudi S. Colorectal cancers with microsatellite instability display mRNA expression signatures characteristic of increased immunogenicity. Mol Cancer 2004; 3:21; http://dx.doi.org/10.1186/1476-4598-3-21; PMID: 15298707
- Strieter RM, Polverini PJ, Arenberg DA, Kunkel SL. The role of CXC chemokines as regulators of angiogenesis. Shock 1995; 4:155 - 60; http://dx.doi.org/10.1097/00024382-199509000-00001; PMID: 8574748
- Padovan E, Spagnoli GC, Ferrantini M, Heberer M. IFN-alpha2a induces IP-10/CXCL10 and MIG/CXCL9 production in monocyte-derived dendritic cells and enhances their capacity to attract and stimulate CD8+ effector T cells. J Leukoc Biol 2002; 71:669 - 76; PMID: 11927654
- Kim HJ, Song DE, Lim SY, Lee SH, Kang JL, Lee SJ, Benveniste EN, Choi YH. Loss of the promyelocytic leukemia protein in gastric cancer: implications for IP-10 expression and tumor-infiltrating lymphocytes. PLoS One 2011; 6:e26264; http://dx.doi.org/10.1371/journal.pone.0026264; PMID: 22022583
- Bandapalli OR, Ehrmann F, Ehemann V, Gaida M, Macher-Goeppinger S, Wente M, Schirmacher P, Brand K. Down-regulation of CXCL1 inhibits tumor growth in colorectal liver metastasis. Cytokine 2012; 57:46 - 53; http://dx.doi.org/10.1016/j.cyto.2011.10.019; PMID: 22129625
- Chiu ST, Hsieh FJ, Chen SW, Chen CL, Shu HF, Li H. Clinicopathologic correlation of up-regulated genes identified using cDNA microarray and real-time reverse transcription-PCR in human colorectal cancer. Cancer Epidemiol Biomarkers Prev 2005; 14:437 - 43; http://dx.doi.org/10.1158/1055-9965.EPI-04-0396; PMID: 15734970
- Jass JR, Atkin WS, Cuzick J, Bussey HJ, Morson BC, Northover JM, Todd IP. The grading of rectal cancer: historical perspectives and a multivariate analysis of 447 cases. Histopathology 1986; 10:437 - 59; http://dx.doi.org/10.1111/j.1365-2559.1986.tb02497.x; PMID: 3721406
- Boissière-Michot F, Denouël A, Boulle N, Guillaume C, Orsetti B, Lopez-Crapez E, Chateau MC, Bibeau F. The non-crosslinking fixative RCL2®-CS100 is compatible with both pathology diagnosis and molecular analyses. Pathol Oncol Res 2013; 19:41 - 53; http://dx.doi.org/10.1007/s12253-012-9556-2; PMID: 22893391
- Chavey C, Bibeau F, Gourgou-Bourgade S, Burlinchon S, Boissière F, Laune D, Roques S, Lazennec G. Oestrogen receptor negative breast cancers exhibit high cytokine content. Breast Cancer Res 2007; 9:R15; http://dx.doi.org/10.1186/bcr1648; PMID: 17261184
