Figures & data
Table 1 List of arsenic minerals in this study, and their formulas
Fig. 1 Digital elevation model of the South Island of New Zealand (downloaded, with permission, from www.geographx.co.nz), showing the topography of the Southern Alps that form the island backbone and induce differing climatic regimes as indicated. Localities mentioned in the text are shown, with the secondary As minerals () indicated. Localities are described in .
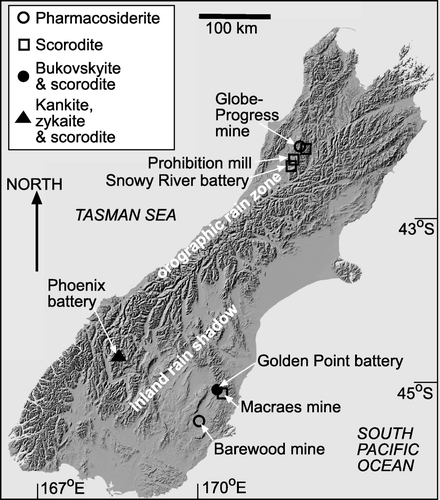
Table 2 Localities mentioned in the text (see ), and their environmental settings
Fig. 2 Ternary diagrams, showing the relative compositions of ideal secondary arsenic minerals in this study (black squares). Broadly related common secondary iron minerals in mine wastes are indicated with open circles. (A) Diagram showing iron, arsenate and sulfate variations. (B) Diagram showing iron arsenate, iron sulfate, and hydration variations.
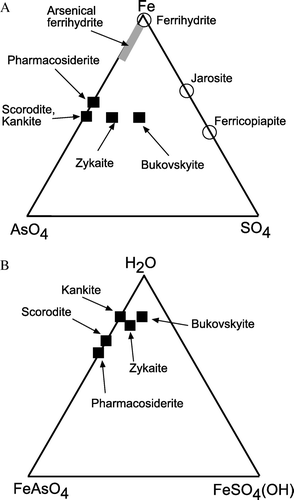
Fig. 3 The concentrations of sodium and potassium in groundwaters and mine waters in the Reefton and Macraes areas, South Island. Mine water compositions are affected by mining and processing activity.
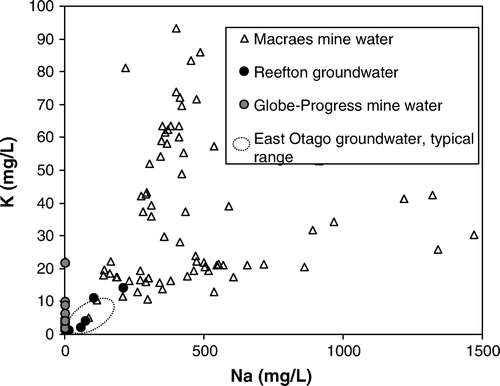
Fig. 4 Electron backscatter images of some secondary arsenic minerals and textures in South Island mine wastes. (A) Crystalline kankite (K) encrustations with scattered quartz (Q) grains, Phoenix battery. (B) Massive fine-grained kankite (K) cementing mine waste grains of quartz (Q) and muscovite (M). (C) Precipitated material from the Globe-Progress mine lower tunnel apron (). Amorphous ferrihydrite (black) and amorphous ferrihydrite with abundant admixed arsenate (Fe,As; middle grey) dominate this material, which is cut by a veinlet of secondary pharmacosiderite (P; pale grey). (D) Scorodite (S; middle grey) forms a cement in mine wastes dominated by dark grey quartz (Q) and muscovite (M). A relict arsenolite grain (white, A) has been partially plucked during specimen preparation, leaving a black hole.

Fig. 5 Compositions of South Island mine waters on a dissolved As versus solution pH diagram. Black triangles are for Otago (east of mountains) (Craw & Pacheco Citation2002). Circles are for mines west of the mountains (DAME database); open circles=gold mines; grey circles=coal mines. Scorodite solubility curves are from Krause & Ettel (Citation1988) and Langmuir et al. (Citation2006).
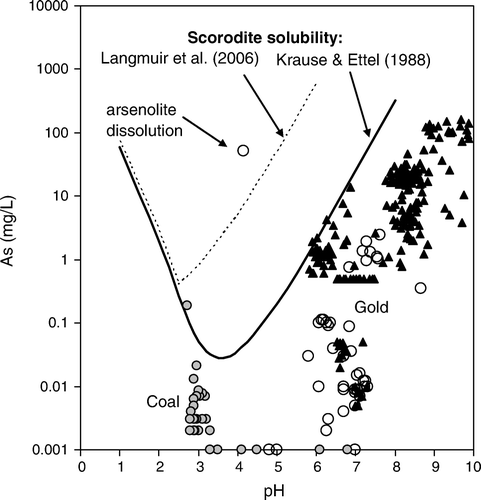
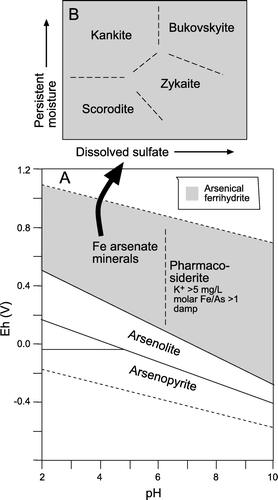
Fig. 6 Relative stabilities of As minerals discussed in this study, as deduced from empirical observations of South Island mine sites. (A) Eh-pH diagram, after Vink (Citation1996) and Craw et al. (Citation2003), for total dissolved As of c. 10-4 moles/L, Fe/As ≥1, and dissolved K+ of c. 5 mg/L. Secondary As minerals are commonly accompanied by arsenical ferrihydrite (grey ornament). The boundary between pharmacosiderite and scorodite-like minerals has not been quantified thermodynamically. (B) Schematic phase diagram showing qualitative controls on iron arsenate mineral formation. The boundaries separating the different minerals in B have not been quantified thermodynamically, but reflect differences in mineral formulae () and observed occurrences (). The ‘persistent moisture’ axis reflects microclimates as outlined in the text.