Figures & data
Figure 1. Flowchart of study population. *Answers to questions regarding TDM of biologic agents were lacking.
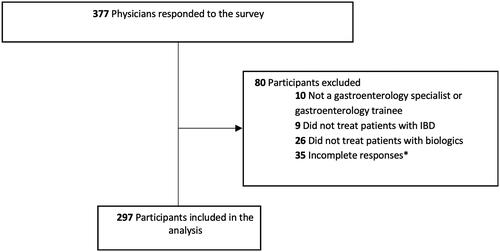
Table 1. Demographic and clinical characteristics of study participants (n = 297).
Table 2. Use of reactive and proactive TDM across different biologic drugs.
Figure 2. The use of reactive versus proactive TDM of biologic agents among physicians in Scandinavia. Distribution of the current use of reactive and proactive TDM in clinical practice in Norway, Denmark, Sweden, Finland, and Iceland presented as frequencies (%). P-values are generated from multivariate logistic regression models, adjusted for country, employment at university hospital, size of hospital catchment area, seniority, and number of IBD contacts per week.
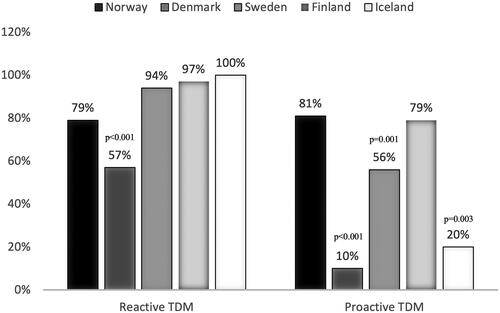
Table 3. Factors associated with use of proactive TDM of biologic agents (n = 297).
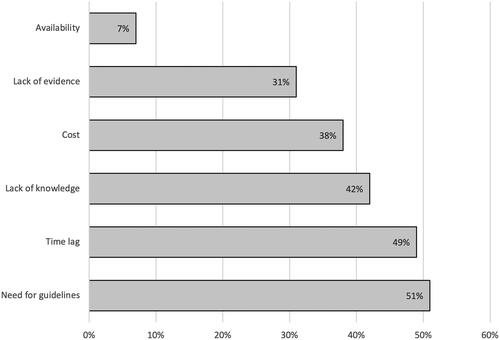
Figure 3. Barriers hindering the implementation of TDM of biological therapy in clinical practice in Scandinavia. Barriers and reasons for not using TDM of biologic agents among physicians in Scandinavia presented as frequencies (%)*. *Missing data: n = 52.
Supplemental Tables
Download PDF (197.1 KB)Data availability statement
The data underlying this article are available in the article and in its online supplementary material.
