Figures & data
Figure 1. Flowchart. AUH: Aarhus University Hospital; AAUH: Aalborg University Hospital; BCSs: breast cancer survivors; AET: adjuvant endocrine therapy; FSFI: Female Sexual Function Index; SCS-W: Sexual Complaint Screener – Women; SD: sexual dysfunction. *At AAUH; the number of women invited to participate was not registered.
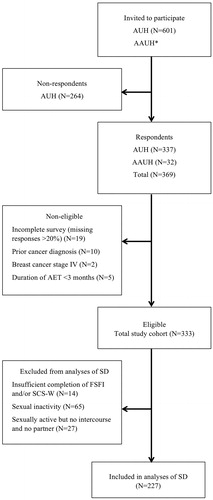
Table 1. Sociodemographic, health-related and clinical characteristics of the total study cohort.
Table 2. Urogenital symptoms prior to the breast cancer diagnosis and at time of study entry.
Table 3. Odds ratios for sexual dysfunction according to selected covariates among sexually active women (N = 227).
Table 4. Adjusted odds ratios for sexual dysfunction according to age, vaginal dryness, psychological well-being, relationship satisfaction and duration of adjuvant endocrine therapy.
