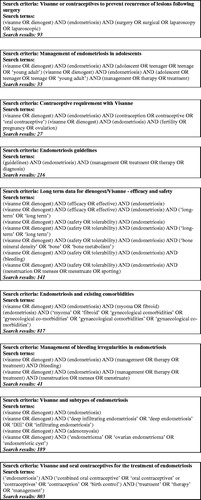
Figures & data
Table 1. Dienogest efficacy and safety studies with a duration ≥1 year.
Table 2. Summary of impact of dienogest treatment on lumbar BMD across studies.
Table 3. Data supporting the efficacy of dienogest for the prevention of postsurgical recurrence.