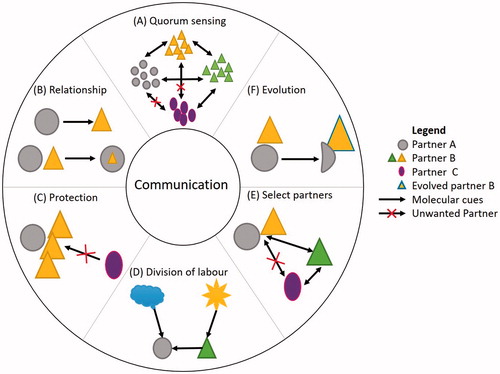
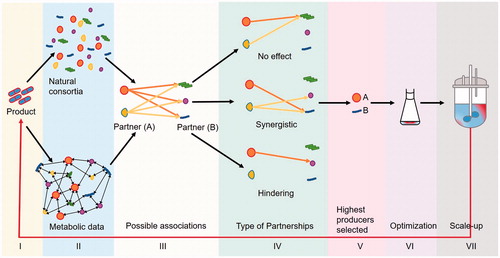
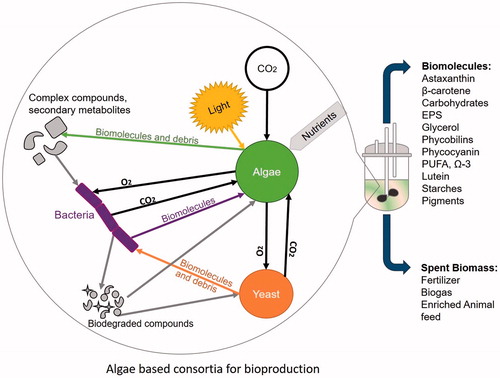
Figures & data
Table 1. Microbial co-cultures in bio-production.
Table 2. A selection of high-value products derived from microalgae species as monocultures.