Figures & data
Figure 1. Metabolites of the isoprenoid biosynthesis pathway and nuclear receptors. The isoprenoid pathway (also known as the mevalonate pathway) produces many isoprenoid-based molecules that can serve as ligands for numerous nuclear receptors. Isoprenoid FPP (farnesyl pyrophosphate) may serve as an endogenous ligand for PPARγ. VDR: vitamin D receptor, CAR: constitutive androstane receptor, ROR: RAR-related orphan receptor, LXR: liver X receptor, PXR: pregnane X receptor, FXR: farnesoid X receptor, SHRs: steroid hormone receptors.
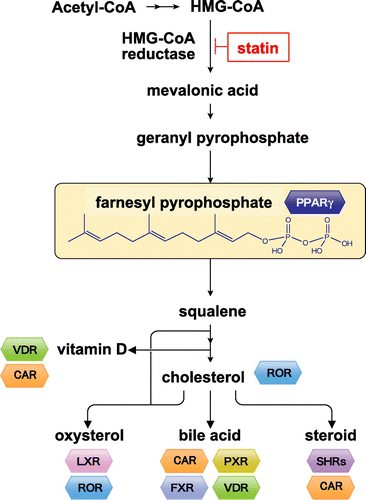
Figure 2. Inflammation of adipose tissue and symptoms of metabolic syndrome. Metabolic syndrome has been found to depend on an inflammatory reaction by the M1 macrophages (Mϕ) that exists in obese white adipose tissue (WAT). M1 Mϕ infiltrate into obese WAT, where they are recruited by MCP-1 from hypertrophied adipocytes, resulting in additional accumulation of M1 Mϕ and an increase of inflammatory cytokines in WAT. These form a positive feedback loop to cause hypertrophy of adipose cells, leading to mutual and chronic inflammation.
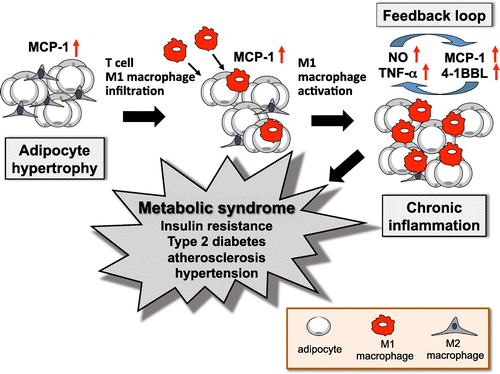
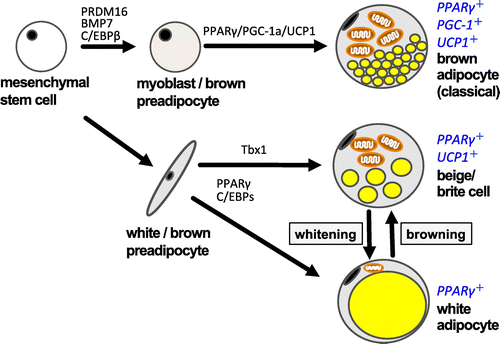
Figure 3. Differentiation scheme of white/brown adipocyte and beige cells. Recently, considerable research has been done on the original stem cell containing UCP-1. Mouse interscapular tissues are rich in brown adipocytes containing UCP-1. This is a classical brown adipocyte, which is generated from the same stem cells as muscle tissues. The brown-like white adipocyte, called the beige cell or brite cell, is reported to be found in white adipose tissue. Recent studies have indicated that beige adipocytes have thermogenic activity comparable to that of classical brown adipocytes and contribute significantly to the regulation of body fat content.