Figures & data
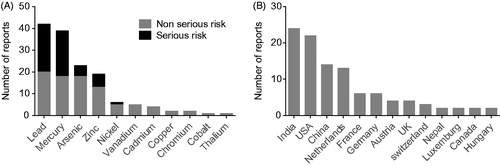
Table 1. Metal contaminants in food supplements. Examples of studies reporting quantifiable levels of arsenic (As), cadmium (Cd), lead (Pb), and mercury (Hg)
Table 2. Examples of studies reporting quantifiable levels of mycotoxins in food supplements
Table 3. Examples of studies reporting quantifiable levels of cyanotoxins in food supplements of marine origin
Table 4. Examples of studies reporting quantifiable levels of pesticides in food supplements
Table 5. Dioxins, PCBs, and PBDEs residues in food supplements.
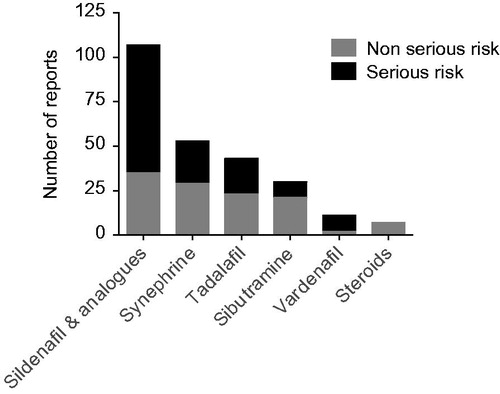
Table 6. Examples of studies reporting adulteration of food supplements with pharmacological active ingredients
Table 7. RASFF reports regarding food supplements from 1998 to 2018.