Figures & data
Figure 1. Particle size distributions directly after precipitation (-□-), after oxidation (-▴-), and after release from the lipid formulation (-⧫-). Indicated values are means ± SD (n ≥ 3).
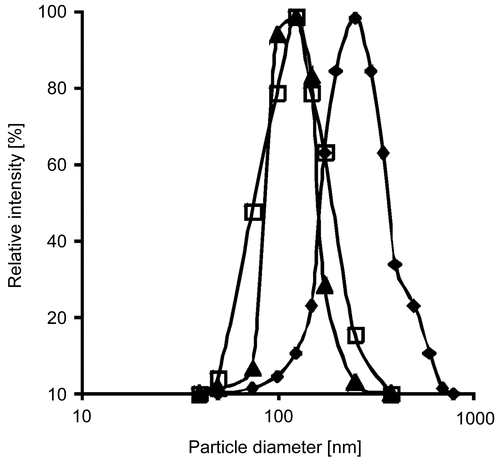
Table 1. Characterization of the utilized PAA100-Cys nanoparticles regarding mean particle diameter, zeta potential, and drug load. Indicated values are means ± SD (n ≥ 3).
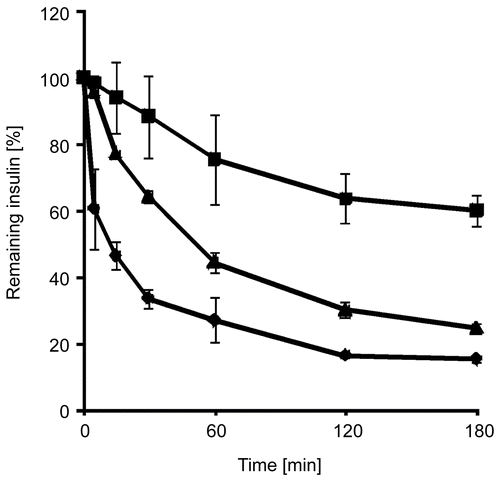
Figure 2. In vitro release of insulin from PAA100-Cys nanoparticles as a percentage of the total amount of insulin present in the utilized formulation. Indicated values are means ± SD (n ≥ 3).
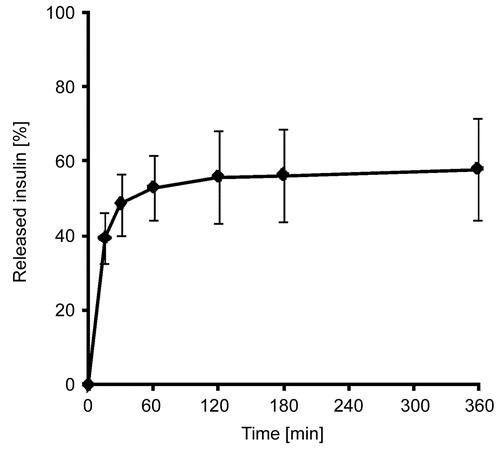
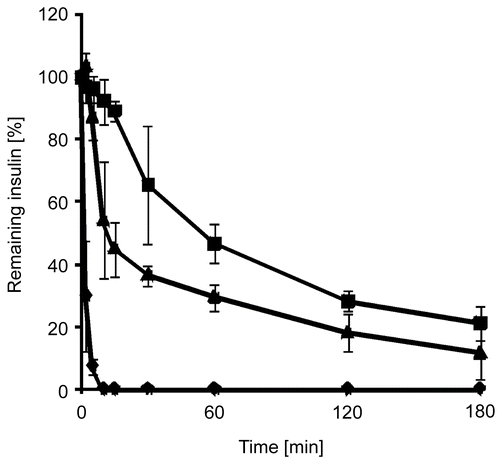
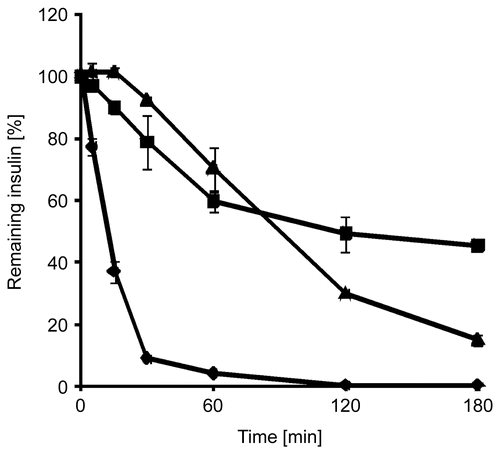
Figure 3. Degradation of insulin by trypsin in an aqueous solution (⧫), as a solid dispersion of insulin in triglyceride (▴), and as a solid dispersion of insulin-loaded PAA100-Cys nanoparticles in triglyceride (▪). Indicated values are means ± SD (n ≥ 3).