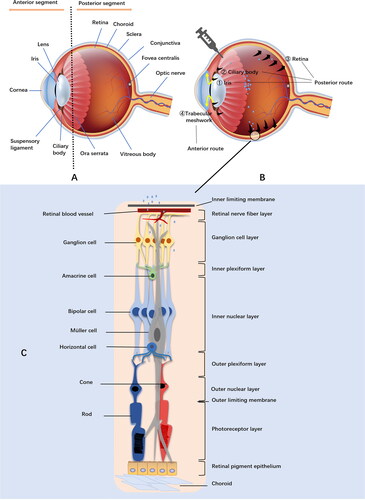
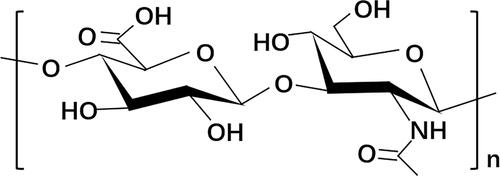
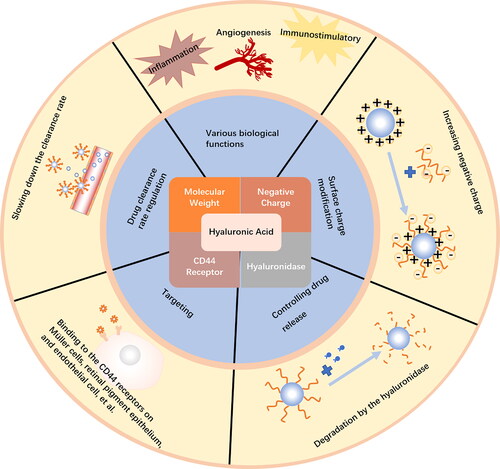
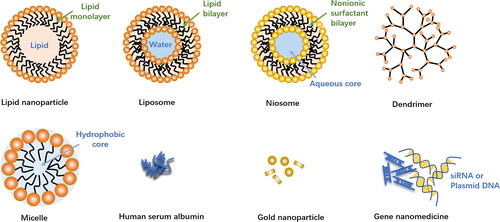
Figures & data
Table 1. Advantages and drawbacks of nanoparticles for intravitreal administration.
Table 2. HA-based nanoparticles.