Figures & data
TABLE 1 Xs and refractive index as a function of °Brix for coffee solutions
TABLE 2 Freezing point of coffee solutions as a function of coffee mass fraction
TABLE 3 Parameters of power law (Eq. 1) for different coffee mass fractions and temperatures
TABLE 4 Viscosity of coffee solutions at different temperatures (T) and coffee mass fractions (Xs) (mPa·s)
TABLE 5 Parameters of Arrhenius equation (Eq. 2) for coffee solutions at different Xs
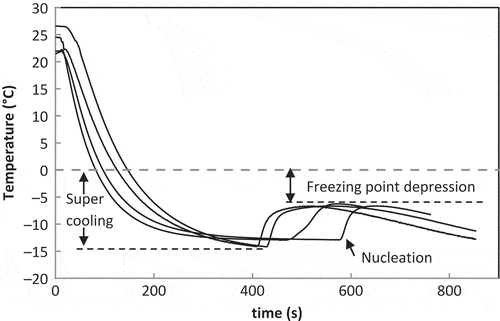
FIGURE 2 Rheogram of coffee solutions at 4°C (□), 2°C (▵), 0°C (◊), −2°C (○), −4°C (×), and −6°C (—). (a) Xs = 0.05, (b) Xs = 0.20, (c) Xs = 0.35, (d) Xs = 0.50. Lines are calculated values using parameters given in .
TABLE 6 Parameters of mathematical models for prediction of coffee solution’s viscosity
FIGURE 3 Coffee solution’s viscosity as a function of temperature and coffee mass fraction. Predicted values using Eq. (11). The curve on surface represents freezing point curve for coffee solutions modeled by Eq. (10).
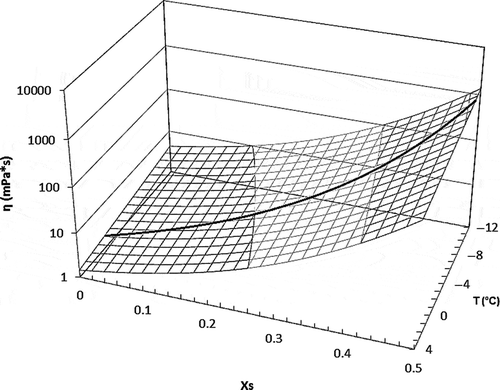
TABLE 7 Density of coffee extract (kg·m−3) as a function of coffee mass fraction and temperature