Figures & data
Figure 2. Complexes of x-substituted tyrosol derivatives (x = NHMe, OMe, and tert-butyl) with BN-nanotube (9, 0).
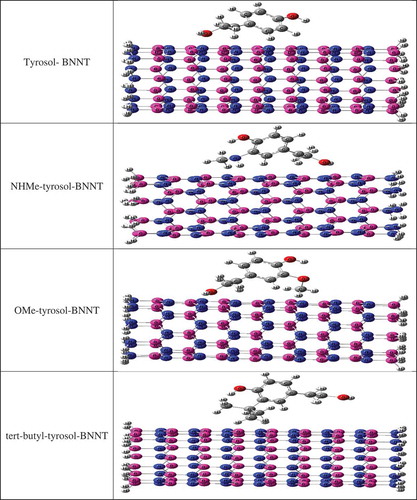
Table 1. The computed BDE and IP values (kJ mol−1) of tyrosol derivatives in the gas phase and water.
Table 2. The computed BDE and IP values (kJ mol−1) of tyrosol derivatives in the benzene, ethanol, and methanol phase.
Table 3. The obtained ∆BDE and ∆IP values of substituted phenols (kJ mol–1) via experimental and theoretical methods.
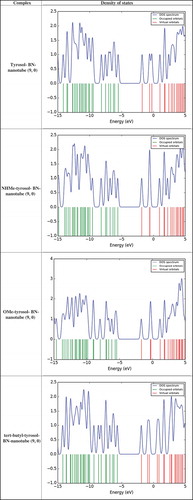
Table 4. The calculated Ead values (kJ mol−1) of tyrosol derivatives on BN-nanotube (9, 0) surface, and the charge transferred (QT (e) in eV) from the tyrosol derivatives to BN-nanotube (9, 0).
Table 5. The calculated Ead values (kJ mol−1) of tyrosol derivatives on BN-nanotube (10, 0), BN-nanotube (8, 0), and BN-nanotube (7, 0) surfaces.