Figures & data
Table 1. Vaccine effectiveness for Settings A and B in the 4Flu model.
Table 2. Annualized disutility due to symptomatic influenza (calculated*).
Table 3. Disutility due to complications (calculated*).
Table 4. The annual clinical outcomes for Settings A and B in the cohort size of 100,000 individuals.
Table 5. The annual health economic outcomes for Settings A and B in the cohort size of 100,000 individuals.
Table 6. Payer perspective scenario analysis annual health economic outcomes for Settings A (egg-adaptation occurs in 100% of all years) and B (egg-adaptation occurs in 55% of all years) in the cohort size of 100,000 individuals.
Table 7. Scenario analysis annual health economic outcomes for Settings A (egg-adaptation occurs in 100% of all years) and B (egg-adaptation occurs in 55% of all years) in the entire German population.
Figure 1. Deterministic sensitivity analysis: impact of the 10 key drivers on the NMB (Settings A [egg-adaptation occurs in 100% of all years] and B [egg-adaptation occurs in 55% of all years], societal perspective, 100,000 individuals). (a) Impact of the 10 key drivers on the NMB for Setting A. (b) Impact of the 10 key drivers on the NMB for Setting B. Abbreviations. aVE, absolute vaccine effectiveness; NMB, net monetary benefit; OSWA, one-way sensitivity analyses; rVE, relative vaccine effectiveness. Setting A: egg-adaptation occurs in 100% of all years. Setting B: egg-adaptation occurs in 55% of all years. % of death and % of hospitalizations are due to the following complications: bronchitis, pneumonia, unspecified upper tract respiratory infection, myocarditis, myocardial infarction, renal, central nervous system, stroke, otitis media, gastrointestinal bleedings where the base case values were changed with ±10%; disutilities due to influenza symptoms, baseline utilities, and disutilities due to influenza hospitalization changed with ±10%.
![Figure 1. Deterministic sensitivity analysis: impact of the 10 key drivers on the NMB (Settings A [egg-adaptation occurs in 100% of all years] and B [egg-adaptation occurs in 55% of all years], societal perspective, 100,000 individuals). (a) Impact of the 10 key drivers on the NMB for Setting A. (b) Impact of the 10 key drivers on the NMB for Setting B. Abbreviations. aVE, absolute vaccine effectiveness; NMB, net monetary benefit; OSWA, one-way sensitivity analyses; rVE, relative vaccine effectiveness. Setting A: egg-adaptation occurs in 100% of all years. Setting B: egg-adaptation occurs in 55% of all years. % of death and % of hospitalizations are due to the following complications: bronchitis, pneumonia, unspecified upper tract respiratory infection, myocarditis, myocardial infarction, renal, central nervous system, stroke, otitis media, gastrointestinal bleedings where the base case values were changed with ±10%; disutilities due to influenza symptoms, baseline utilities, and disutilities due to influenza hospitalization changed with ±10%.](/cms/asset/9415ea3e-865e-40de-afe4-badd83d4e842/ijme_a_1908000_f0001_c.jpg)
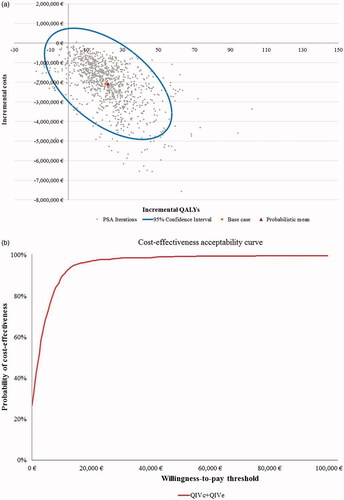
Figure 2. Scatter plot and cost-effectiveness acceptability curve for PSA with 1,000 iterations for Setting A (egg-adaptation occurs in 100% of years), societal perspective. (a) Scatter plot of PSA for Setting A. (b) Cost-effectiveness acceptability curve (CEAC) for Setting A. Abbreviations. CEAC, cost-effectiveness acceptability curve; PSA, probabilistic sensitivity analyses; QALYs, quality-adjusted life-years; QIVc, cell-based quadrivalent influenza vaccine; QIVe, egg-based quadrivalent influenza vaccine; €, Euros. Setting A: egg-adaptation occurs in 100% of all years.
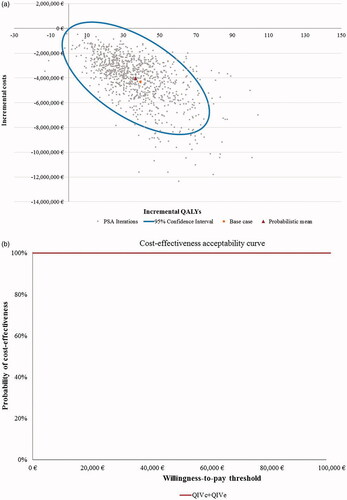
Figure 3. Scatter plot and cost-effectiveness acceptability curve for PSA with 1,000 iterations for Setting B (egg-adaptation occurs in 55% of years), societal perspective. (a) Scatter plot of PSA for Setting B. (b) Cost-effectiveness acceptability curve (CEAC) for Setting B. Abbreviations. CEAC, cost-effectiveness acceptability curve; PSA, probabilistic sensitivity analyses; QALYs, quality-adjusted life-years; QIVc, cell-based quadrivalent influenza vaccine; QIVe, egg-based quadrivalent influenza vaccine; €, Euros. Setting B: egg-adaptation occurs in 55% of all years.