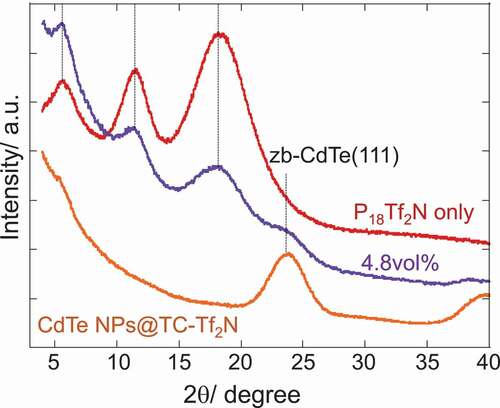
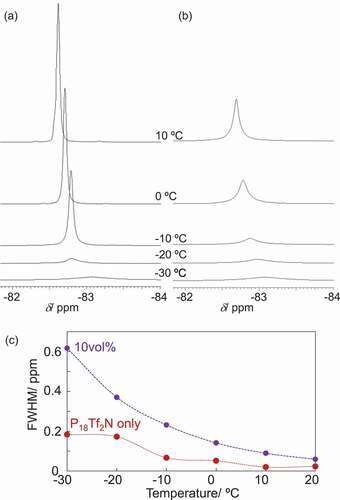
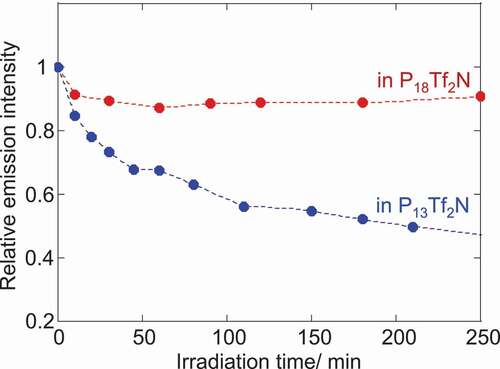
Figures & data

Figure 1. DSC thermograms of IL-CdTe NP composites with (a) P12-, (b) P14- and (c) P18Tf2N together with those of 1-alkyl-1-pyrrolidinium salts without NPs
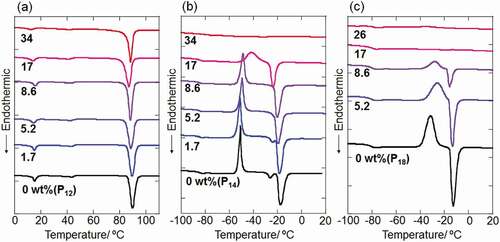
Figure 2. Plots of relative melting enthalpy of 1-alkyl-1-methylpyrrolidinium Tf2 N salts as a function of NP-volume fraction. ΔH0 corresponds to the melting enthalpy of ILs without NPs
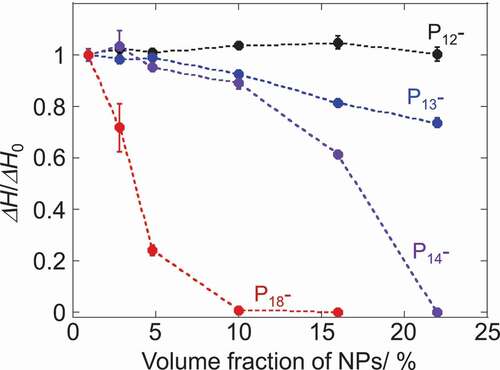
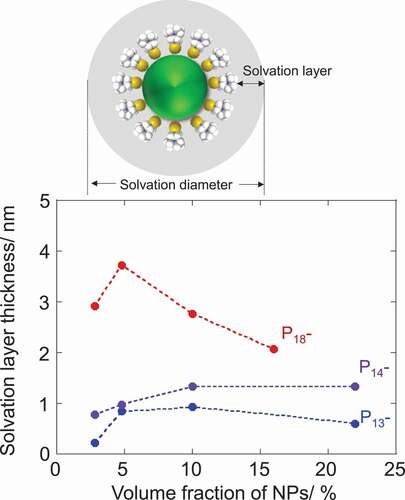
Figure 3. Plots of estimated solvation layer thickness on NPs in 1-alkyl-1-methylpyrrolidinium Tf2 N salts as a function of NP-volume fraction together with a schematic representation of solvation layer (above)