Figures & data
Figure 1. Thymic T-cell lymphoma cells from Lck-Dlx5 mice have elevated Akt activity and elevated Myc expression. Proteins from 3 lymphoma cell lines (F86-786, F86-793 and F86-801) derived from Lck-Dlx5 mice and normal thymocytes from 2 wild type (WT) littermates were analyzed by immunoblotting. Expression of Myc-tagged Dlx5, Myc, phospho-Akt (ser473), total Akt and Pten were evaluated. (A) Note that activation of Akt in T-cell lymphoma cells was associated with loss of Pten expression. Array-CGH profiles of 3 Lxk-Dlx5 lymphoma cell lines (including 2 matching those shown in panel A, F47-0 showed the similar pattern on a separate gel) with recurrent trisomy of chromosome (Chr) 15, location of Myc, in tumor cells (B).
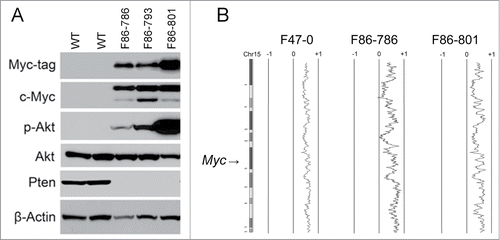
Table 1. Pten mutations in T-cell lymphoma lines from Lck-Dlx5 mice
Figure 2. Low concentrations of BEZ235 induce dose-dependent cell cycle inhibition and decreased cell viability. Three different Lck-Dlx5 lymphoma cell lines were treated with BEZ235 at the indicated concentrations. Cell viability/proliferation were monitored by MTS assay (A). Cell cycle analysis was performed by flow cytometry. Histograms summarizing results from 3 independent flow cytometry experiments are shown in panels (B). Western blot analysis of phospho-Akt, phospho-S6, cyclin A, cyclin D1 and β-actin (C). Higher concentrations of BEZ235 (≥0.5 µM) treatment for 24 h cause apoptosis in these lymphoma cells as shown by an increase in sub-G1 cells (D), and activation of caspase 3 (E).
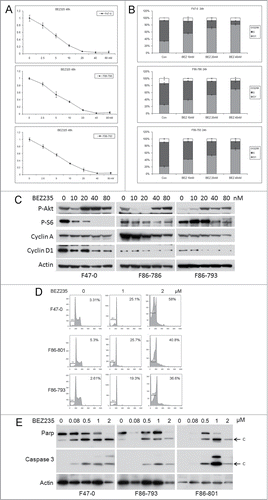
Figure 3. Bromodomain inhibitor JQ1 induces dose-dependent cell cycle inhibition and reduced cell viability in lymphoma cells from Lck-Dlx5 mice. Cell lines were treated with JQ1 at the indicated concentrations. Cell viability/proliferation was monitored by MTS assay (A). Cell cycle analysis was performed by flow cytometry (B). Western blot analysis depicting expression of Myc, cyclin A, cyclin D1 and β-actin (C).
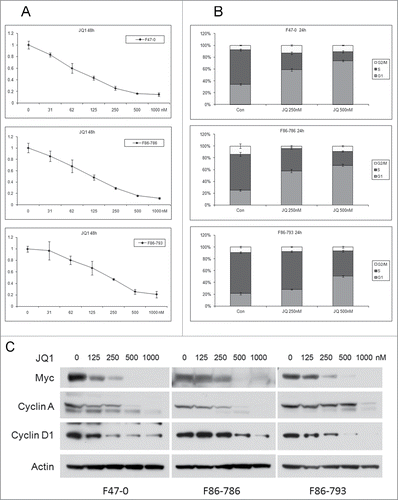
Figure 4. Low concentrations of BEZ235 and JQ1 cooperate to induce enhanced cell cycle arrest. Lck-Dlx5 lymphoma lines were treated with BEZ235 and JQ1 separately or in combination at the indicated concentrations. Cell viability/proliferation was monitored by MTS assay (A, B), and cell cycle analysis was performed by flow cytometry (C, D). BEZ235 (B, 40 nM) or RAD001 (R, 1 nM), when combined with JQ1 (J, 250 nM) for 24 h, cooperatively suppress expression of cyclin A, cyclin D1 and phospho-Cdk6 (E).
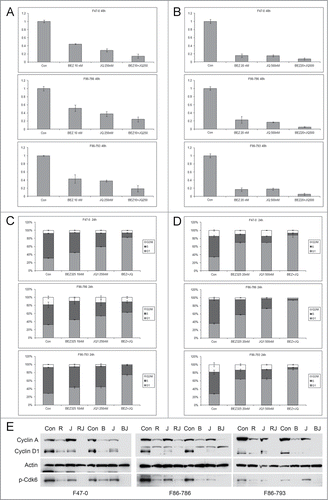
Figure 5. The specific Myc inhibitor 10058-F4 induces apoptosis in lymphoma cells from Lck-Dlx5 mice and cooperates with BEZ235 to enhance apoptosis in these cells). 10058-F4 along triggers apoptotic sub-G1 DNA condensation (A). 10058-F4 at 20 µM potently promotes apoptosis in Lck-Dlx5 lymphoma cells as early as 12 h after initiating treatment, and 10058-F4 treatment results in cleavage of Parp and caspase 3 as well as decreased expression of Myc in a dose-dependent manner (B). 10 µM 10058-F4 (F4) cooperates with BEZ235 (0.5 µM) in triggering apoptotic sub-G1 DNA condensation (C) as well as Parp cleavage and decreased Myc protein levels (D).
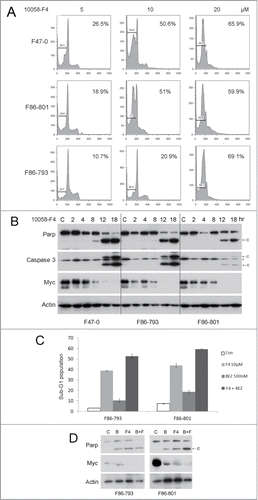
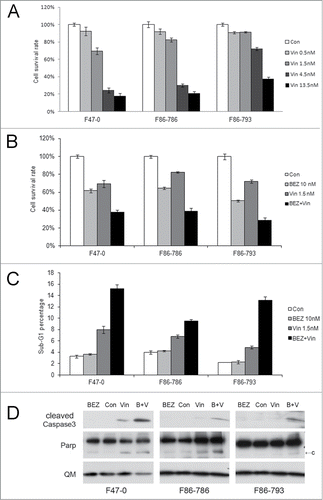
Figure 6. Apoptosis induction in Lck-Dlx5 lymphoma cells by vincristine and BEZ235. Vincristine-induced apoptosis is dose-dependent (A). Cells were treated with vincristine (Vin) at the indicated concentrations for 24 h, and cell viability/proliferation was monitored by MTS assay. mTORC1 inhibition potentiates vincristine-induced apoptosis (B). Cells were treated with 10 nM BEZ235 (BEZ) separately or in combination with 1.5 nM vincristine for 24 h, and MTS assay was performed. Apoptotic sub-G1 cell populations were analyzed by using flow cytometry (C). Western blot analysis depicting activation of caspase 3 (D).