Figures & data
Figure 1. Reduced Tor activity in Vps16A mutants. (A) Representative western blots show that the levels of lipidated, autophagosome-associated Atg8a-II are much higher in well-fed Vps16A mutants than in control or Syx17-mutant L3-stage larvae. (B) The levels of P-S6k/Phospho-RPS6KB1 (Thr389) are reduced, while the ratio of hyperphosphorylated (slower migrating) to hypophosphorylated (faster migrating) Atg13 is increased in Vps16A mutants, compared to well-fed control or Syx17-mutant larvae. Asterisk marks a nonspecific band. (C) The level of P-Thor/Phospho-EIF4EBP1 (Thr37/46) is decreased in GFP-marked Vps16A-null mutant cells compared to neighboring control cells in mosaic fat bodies of well-fed larvae. (D) Venus-tagged raptor displays punctate localization in fat body cells of well-fed wild-type larvae, and colocalizes with Cp1 (Cathepsin L)-positive lysosomes (arrowheads). (E) Punctate localization of Venus-raptor is lost in Vps16A mutants. (F) RT-PCR analysis reveals the obvious transcriptional upregulation of Atg8a and ref(2)P/p62 in Vps16A mutants when compared to control or Syx17-mutant animals. Overexpression of Rheb in Vps16A mutants restores Atg8a and ref(2)P/p62 mRNA levels to those seen in control animals. Scale bar equals 20 µm for panels (C-E). Numbers represent protein level ratios estimated by densitometry in panels (A and B).
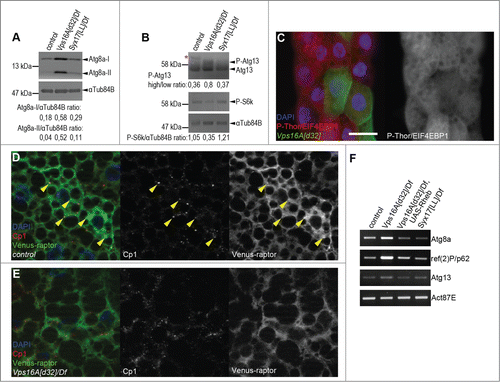
Figure 2. Reactivation of Tor suppresses autophagosome formation and restores growth and developmental timing in Vps16A mutants. (A) Overexpression of Rheb in GFP-positive cell clones suppresses endogenous Atg8a-positive autophagosome formation in starved Vps16A-mutant fat body cells. (B) Ultrastructural analysis shows the large-scale accumulation of double-membrane autophagosomes (arrowheads) in fat body cells of well-fed Vps16A-mutant larvae. (C and C') Fat body-specific expression of Rheb driven by cg-Gal4 suppresses autophagosome formation in fat cells (C) but not in the tracheal epithel (C') of well-fed Vps16A-mutant larvae. (D) The size defect of Vps16A-mutant larvae 4 d after egg laying is rescued by low-level expression of Rheb. (E) Low-level expression of Rheb using an uninduced hs-Gal4 driver rescues developmental timing, as assessed by the ratio of wandering L3-stage larvae 5 d after egg laying, compared to heterozygous siblings. N=15 and N=12 culture vials were evaluated to calculate data for Vps16A mutants and Rheb-expressing Vps16A mutants, respectively, with an average of 30 larvae counted per vial. Note that the expected Mendelian ratio is 33%. Scale bars :20 µm (A), and 1 µm (B, C, C').
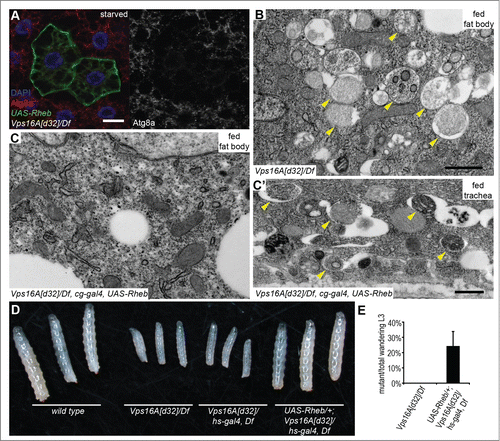
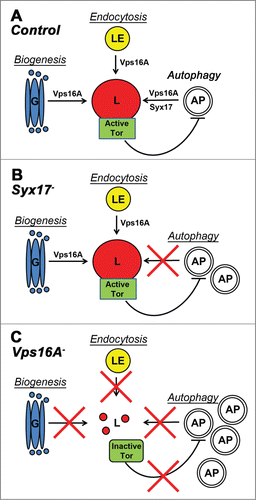
Figure 3. A model of Vps16A function. (A) Vps16A is required for multiple transport routes to lysosomes, including their biogenesis, endocytosis, and autophagy. (B) Loss of Syx17 blocks autophagosome-lysosome fusion and autophagic flux without affecting lysosome-dependent Tor activation. (C) Loss of Vps16A inhibits all 3 transport routes to lysosomes, which results in decreased Tor activity and increased autophagosome formation, in addition to a block of autophagosome-lysosome fusion. AP, autophagosome; G, Golgi; L, lysosome; LE, late endosome.