Figures & data
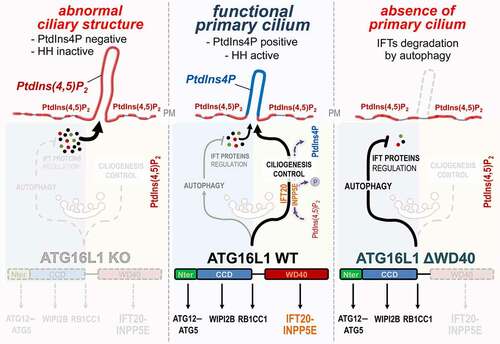
Figure 1. ATG16L1 at the crossroad of autophagy and ciliogenesis regulation. Center: In response to serum deprivation, ATG16L1 participates in IFT proteins turnover by promoting their autophagic degradation, notably via its N-terminal and coiled coil (CCD) domains. Conversely, ATG16L1 enables the proper trafficking of IFT20, via its WD40 domain, as well as the PtdIns(4,5)P2-associated phosphatase INPP5E at the site of ciliogenesis. This allows the proper conversion of PtdIns(4,5)P2 into PtdIns4P which accumulates in the axoneme of the PC. Right: In contrast, the ΔWD40-ATG16L1 mutant is not able to drive IFT20 and INPP5E to the plasma membrane, but autophagy capacities of ATG16L1 are not affected, thus resulting in massive degradation of IFT proteins and abolition of ciliogenesis. Left: in the absence of ATG16L1, autophagy is inhibited, IFT proteins accumulate and INPP5E is not targeted to the plasma membrane. Consequently, an abnormal ciliary-like structure is observed which is devoided of PtdIns4P and key functional markers like the Hedgehog pathway (HH) signaling components