Figures & data
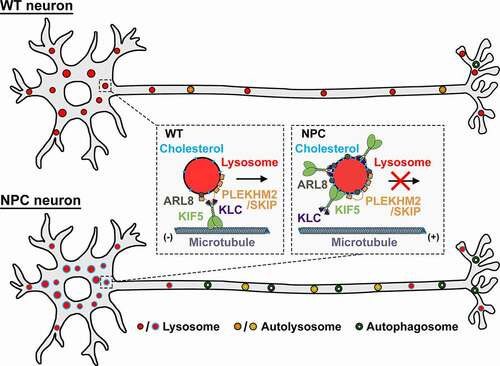
Figure 1. Schematic model showing lipid-mediated sequestration of motor-adaptor proteins impairs axonal lysosome delivery, contributing to autophagic stress in NPC axons. In WT neurons, the ARL8-PLEKHM2/SKIP-kinesin-1 complex is appropriately recruited to and assembled on lysosome membranes in the soma, driving lysosome transport into axons to facilitate axonal autophagosome maturation and clearance. In NPC neurons, altered membrane lipid composition on somatic lysosomes sequesters ARL8 and kinesin-1 independent of PLEKHM2/SKIP, disrupting lysosome transport to distal axons. Inefficient lysosome delivery impairs axonal autophagosome maturation, resulting in increased autophagic stress in presymptomatic NPC axons. Reducing lysosomal membrane cholesterol with HPCD treatment releases ARL8 and kinesin-1 sequestration, thus rescuing lysosome transport into axons and reducing axonal autophagosome accumulation at early stages of NPC disease