Figures & data
Table 1. Percent inhibition of trypsin and chymotrypsin activity.
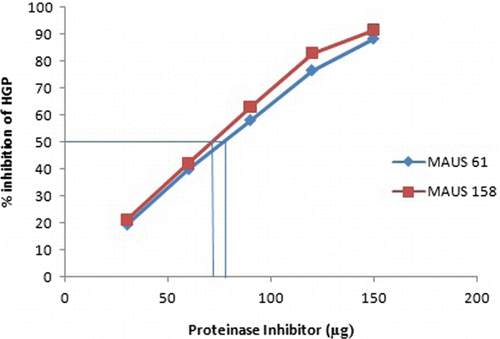
Table 2. Percent inhibition of HGP by crude and F1 fraction.
Table 3. Specific activity and yield of proteinase inhibitors after each step of partial purification.
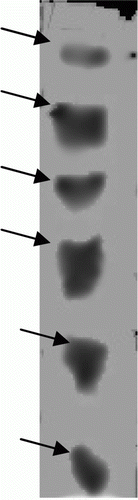
Figure 3. Isoforms of specific trypsin and chymotrypsin proteases using overlay technique. (a) Trypsin like proteases (using BApNA substrate) and (b) chymotrypsin like proteases (using SAALpNA substrate).

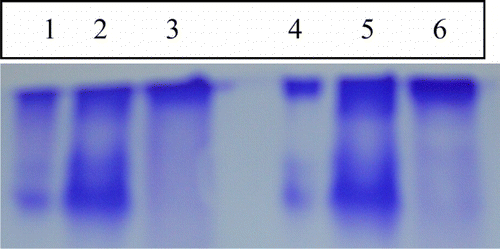
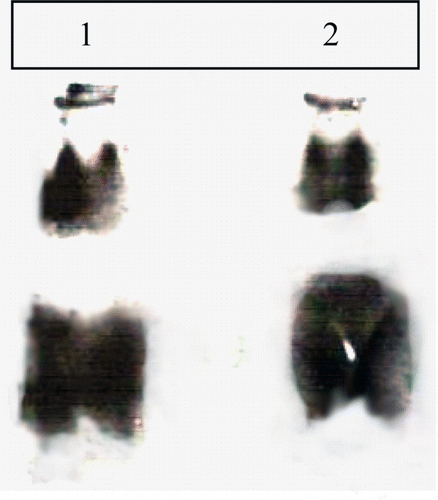
Figure 4. (a) Gel–X-ray film contact technique and (b) zymography. Lanes 1 and 3 corresponds to the F1 and F2 saturated fraction of soybean cultivar MAUS-158. Lanes 2 and 4 corresponds to the F1 and F2 saturated fraction of soybean cultivar MAUS-61.