Figures & data
Table 1. Summary characteristics of baseline demographic and chemistry biomarker covariates among participants who received IV or SC VRC01 in HVTN104 (n = 84).
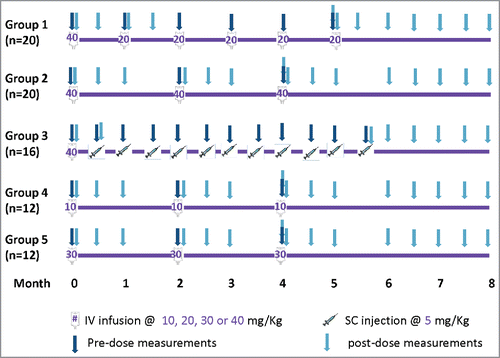
Figure 2. Individual-level concentration (log-scale)-time panel plots for VRC01 recipients in Groups 1–5 (Panels A-E) of HVTN104. Panel A – E display data from Groups 1, 2, 4, 5, and 3, respectively. “+” indicates no product administrations, an open circle indicates product administration, and a filled circle indicates a missed product administration.
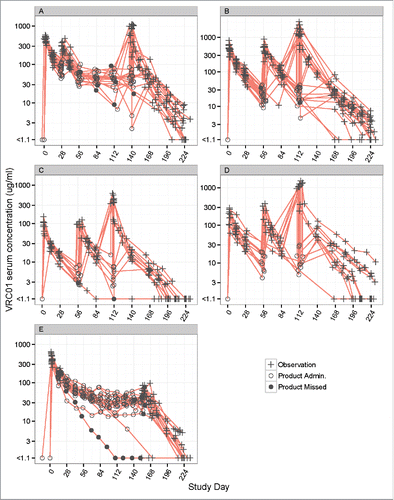
Table 2. popPK parameter estimates of VRC01 based on the modeling of all IV infusion and SC injection groups in HVTN104. %RSE: relative standard error of the fixed effects estimate, calculated as (SE/Estimate) * 100. %CV: coefficient of variation for random effect estimates, calculated as * 100. R: correlation coefficient between random effect estimates.
Figure 3. Goodness-of-fit plots obtained from the final model of IV infusion (red) and SC injection (blue) groups in HVTN104. Panels A-C are model diagnostic plots: (A) observed vs. individual-predicted concentration with an identity line, (B) conditional weighted residual vs. population predicted concentration, and (C) conditional weighted residual vs. time since first IV or SC dose (days). Data from multiple time-points of a participant are individually displayed, not aggregated. Panels D-F are visual prediction check (VPC) plots: (D) dose-normalized observed data from both the q4 and q8 weekly groups and simulated data after a single infusion of 10, 20, 30, or 40 mg/kg IV VRC01, (E) dose-normalized observed data from the SC injection Group 3 with an IV loading dose, and (F) dose-normalized observed data from the q8 weekly groups and simulated data after multiple infusions of 10, 30, or 40 mg/kg IV VRC01. In the VPC plots, lines show medians, and shaded areas show bands covered by the 2.5th and 97.5th percentiles for simulated concentrations. Simulated concentrations were computed from 1000 trials simulated using dosing, sampling, and covariate values from the observed data set.
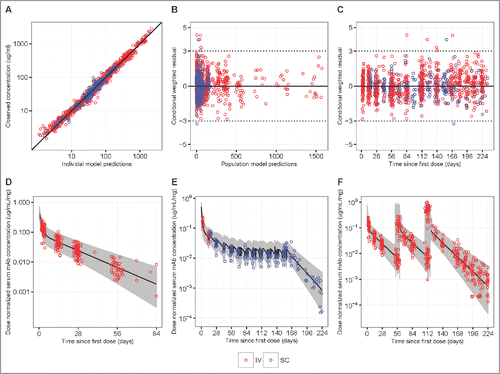
Figure 4. Observed vs. individual-predicted concentrations with an identify line in VRC602 based on the final HVTN 104 model for participants in the IV infusion (red) and SC injection (blue) groups.
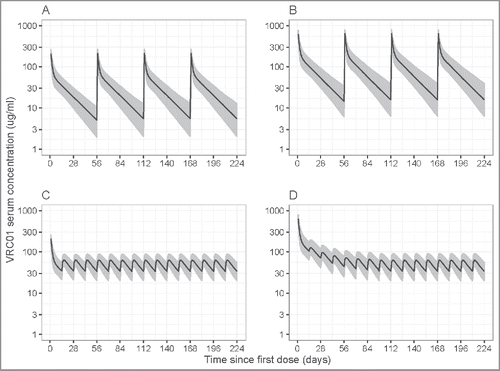
Figure 5. Simulated VRC01 serum concentrations after steady-state is reached under different regimens of multiple-dose IV or SC VRC01. Panels A and B are for the 8-weekly 10 mg/kg and 30 mg/kg IV VRC01 regimens, respectively; Panels C and D are for the 2-weekly 5 mg/kg SC VRC01 regimens, with an IV loading dose of 10 mg/kg and 30 mg/kg, respectively. Lines show medians, and shaded areas show bands covered by the 2.5th and 97.5th percentiles for simulated concentrations. Simulated concentrations were computed from 1000 trials simulated using weight = 74.5 kg.