Figures & data
Table 1. Summary of E. dermatitidis caused infections case reports since 2007.
Table 2. Virulence factors of Exophiala dermatitidis.
Table 3. MIC ranges of several drugs (No. of isolates) against E. dermatitidis isolates from various origins. AMB = amphotericin B, ANI = anidulafungin, CAS = caspofungin, COL = colistin, FLC = fluconazole, ISC = isavuconazole, ITC = itraconazole, MICA = micafungin, POS = posaconazole, TBF = terbinafine, VRC = voriconazole, 5-FC = fluorcytosine.
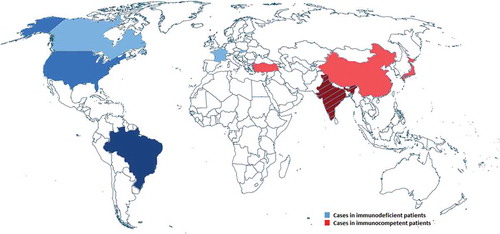
![Figure 2. Number of cases of E. dermatitidis infections reported in the literature from 1960 to May 2018. Data until 1999 were retrieved from Taj-Aldeen et al. [Citation12]. Latest reports were recorded by carefully scrutinized for case reports.](/cms/asset/f5fc1e19-7d12-4c7e-96ed-6de12ec66cce/kvir_a_1596504_f0002_b.gif)
![Figure 4. Survival curve of Galleria mellonella infected with E. dermatitidis Environmental isolates (E), CF patients isolates (CF) and Isolates from Asian, immunocompetent patients (PA). Depicted values are means of nine tested strains. Each strain was tested in triplicate. Taken from: Olsowski et al., 2018 [Citation91].](/cms/asset/6f12a4a9-9016-429a-b76e-78ad52b45f40/kvir_a_1596504_f0004_b.gif)
![Figure 5. E. dermatitidis (CBS 116372) biofilm after 48 h of incubation at 35°C, fixation in methanol and staining by acridine orange in a confocal laser scan microscopy. Taken from: Kirchhoff et al., 2017 [Citation130].](/cms/asset/d9d1b8e9-a2fd-402a-920b-a2664f40eac3/kvir_a_1596504_f0005_oc.jpg)
