Figures & data
Table 1. Residual transmission risk in mothers with undetectable viral load at delivery based on the timing of treatment initiation French Perinatal Study (2000-2011)
Table 2. General principles regarding the use of antiretroviral (ARV) drugs during pregnancy
Figure 1. Evolution of the risk of transmission to the child depending on the intensity of antiretroviral treatment in mono, bi, or triple therapy. French Perinatal Study 1985-2017 (unpublished). NRTI: nucleos(t)ide reverserse transcriptase inhibitor
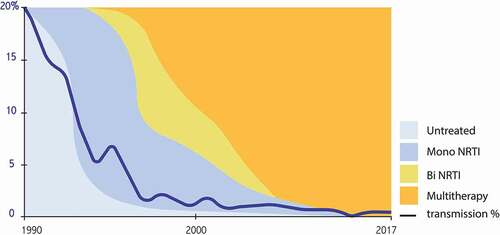
Figure 2. Theoretical cascade of “missed opportunities” for screening and treatment of pregnant women in low-resource countries
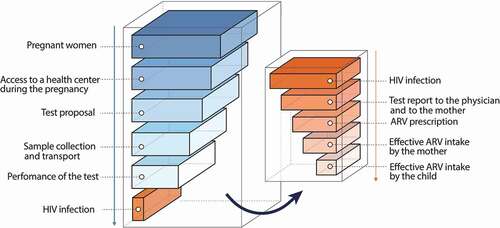
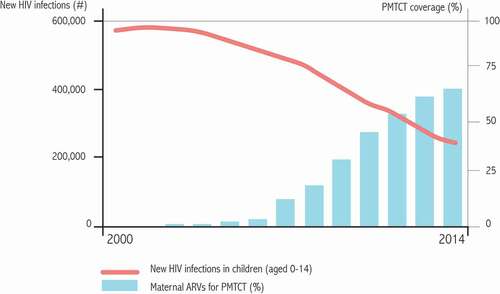
Figure 3. Evolution of the proportion of pregnant women tested for HIV-1 in 12 selected countries and evolution of the number of infected children