Figures & data
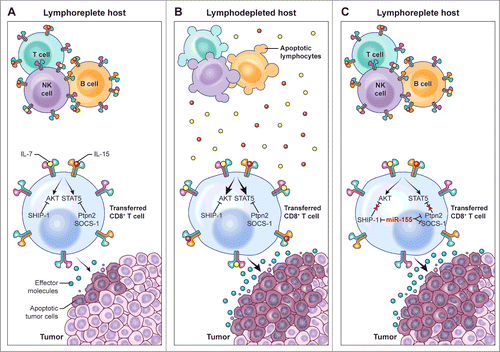
Figure 1. miR-155 augments CD8+ T cell antitumor activity in lymphoreplete hosts by enhancing responsiveness to limited amounts of homeostatic γC cytokines. (A) In the lymphoreplete host, homeostatic cytokine signaling, which is essential to sustain the antitumor function of adoptively transferred tumor-specific CD8+ T cells, is limited by the activity of multiple negative regulators such as SHIP-1, SOCS-1, and Ptpn2. (B) Removal of cytokine “sinks” by lymphodepletion increases the availability of homeostatic γC cytokines, overcoming the inhibitory effects of SHIP-1, SOCS-1, and Ptpn2, thus resulting in enhanced CD8+ T cell antitumor responses. (C) Overexpression of miR-155 in adoptively transferred tumor-specific CD8+ T cells inhibits the expression of SHIP-1, SOCS-1, and Ptpn2, thus resulting in enhanced cytokine signaling and antitumor responses in the presence of limited amounts of homeostatic γC cytokines available in lymphoreplete hosts.