Figures & data
Figure 1. Overview of cancer types and tumor-associated antigens (TAAs) in published and ongoing clinical trials testing DC-based vaccination in cancer patients. A survey of the PubMed database (for published clinical trials; left) or Clinicaltrials.gov database (for ongoing clinical trials; right) concerning the use of DC-based vaccines in cancer patients is presented relative to (1) cancer type; (2) source of antigens; and (3) specific TAAs (when relevant). Only entities applicable to ≥ 2 clinical trials (published studies) or ≥ 3 clinical trials (ongoing studies) are shown. DC, dendritic cell; GBM, glioblastoma; HGG, high-grade glioma; RCC, renal cell carcinoma; TAA, tumor-associated antigens.
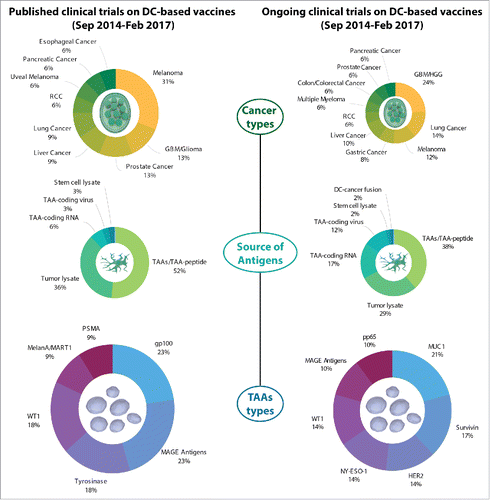
Table 1. Clinical trials currently testing the therapeutic efficacy of dendritic cell-based anticancer immunotherapy.
