Figures & data
Table 1. Patient characteristics and clinical response.
Table 2. GPC3 specific CTL response.
Figure 1. The plasma and serum levels of 3 tumor markers—GPC3 (pg/mL), AFP (mg/mL), and PIVKA-II (AU/mL)—in 11 patients during follow-up. The cut-off thresholds for AFP and PIVKA-II were 10 ng/mL and 40 mAU/mL, respectively.
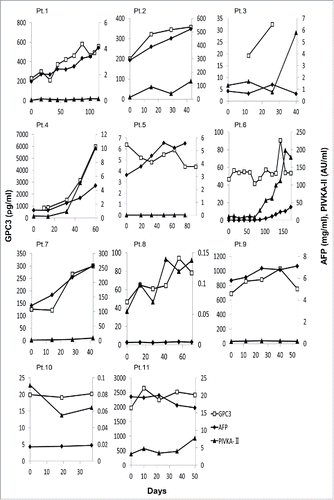
Figure 2. Skin reactions at the injection site in patients after third vaccination. The numbers within the parentheses represent the maximum spot number of GPC3-specific CTLs after vaccination. The 2 patients on the left exhibited stronger injection site reactions relative to those shown on the right.
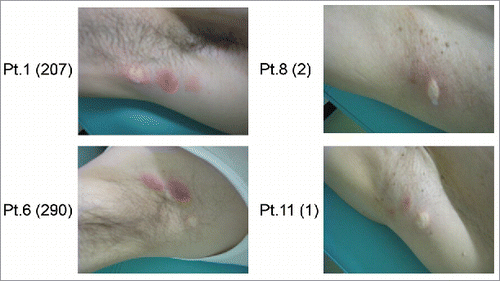
Figure 3. GPC3 peptide vaccine improved overall survivals correlated with peptide-specific CTLs. (A) Kaplan-Meier curves for overall survival. Patients with GPC3-specific CTL frequencies ≥ 50 exhibited longer survival than those with GPC3-specfic CTL frequencies < 50 (p = 0.178). (B) Correlation between GPC3-specfic CTL frequencies and overall survival. GPC3-specfic CTL frequencies after vaccination were significantly correlated with overall survival (p = 0.032, r = 0.645).
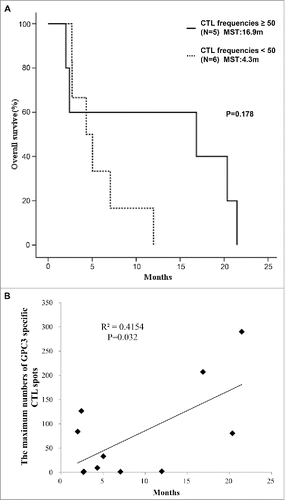
Figure 4. Immunological monitoring of GPC3 peptide-specific T cell responses (A) Ex vivo IFN-γ ELISPOT assay for GPC3 in 5 × 105 PBMCs was performed before and after vaccination in Patient 6. The spot number indicates the number of GPC3 peptide-specific CTLs. The number of IFN-γ-positive spots increased from 0 to 290 in wells preincubated with GPC3 peptide. (B) Ex vivo GPC3 Dextramer staining before and after vaccination in Patient 6. GPC3 peptide-specific CTL frequency is indicated as the percentage of Dextramer-positive CTLs to CD8-positive cells in PBMCs and tumor specimens. (C) Establishment of GPC3 peptide-specific CTL clones in the tumor specimen. Dextramer analysis (left) and IFN-γ ELISPOT assay (right) of the established clones are shown. (D) CTL clone reactivity (TIL 1) against cancer cell lines. Cytotoxic effects of CTL clones against peptide-pulsed T2A24 target cells. HIV583–591 peptide-pulsed targets were used as negative controls. (E) IFN-γ ELISPOT assay against SK-Hep-1/vec, SK-Hep-1/hGPC3, and peptide-pulsed T2A24. Effector/target (E/T) ratio = 0.2. (F-G) Cytokine production by CTL clones (1.0 × 105 cells/well) after 24-h co-culture with the indicated target cells (5 × 104 cells/well). Data represent mean ± SD of triplicate cultures.
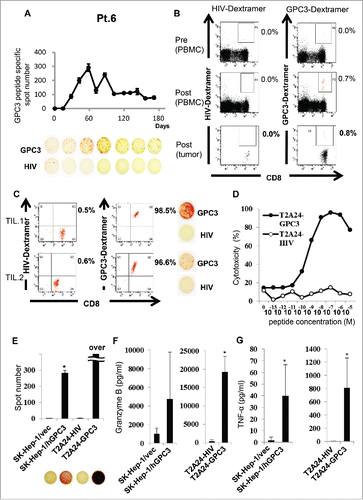
Table 3. Sequence analysis of GPC3 specific CTLs sorted from PBMCs.
