Figures & data
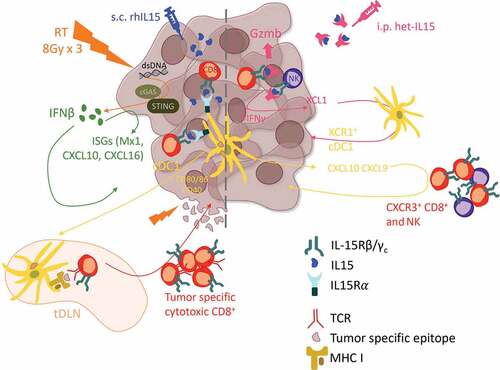
Figure 1. Proposed model of the interactions between radiation and IL15 in the tumor microenvironment promoting anti-tumor immunity. (Left) In advanced tumors optimized radiation therapy regimens elicit cancer-intrinsic activation of the type I IFN pathway and tumor antigen exposure promoting the recruitment of cDC1 that cross-present tumor antigens and transpresent the subcutaneously administered recombinant IL15, resulting in cytotoxic CD8+ T cell infiltration, tumor control and establishment of anti-tumor effector and memory responses. (Right) In more immunogenic/less advanced tumors systemic treatment with biologically active IL15-IL15Rα dimers (het-IL15) expands and activates preexisting anti-tumor CD8+ T and NK cell, which infiltrate the tumor and secrete XCL1 to recruit cDC1. cDC1, in turn, promote further tumor infiltration by CD8+ T and NK cells.