Figures & data
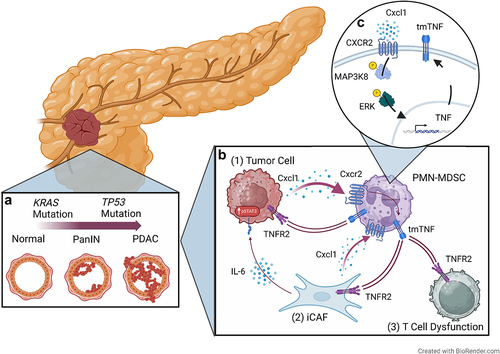
Figure 1. Transmembrane TNF-TNFR2 signaling as a Critical Immunoregulatory Node in pancreatic cancer. leveraging KRAS-TP53 co-altered PDAC as a model for high-risk biology (inset A), our manuscript in Cancer Discovery reveals how unifying cancer cell-autonomous mechanisms, e.g., Creb activation, regulate pro-inflammatory chemokines such as Cxcl1 to dictate functional CXCR2+ neutrophilic myeloid-derived suppressor cell (PMN-MDSC) plasticity and enforce T-cell exclusion in the PDAC tumor microenvironment (inset B). Cxcl1-CXCR2 engagement regulates tumor necrosis factor (TNF) production via a MAP kinase-dependent mechanism in PMN-MDSCs (inset C). Via transmembrane (tm)TNF-TNFR2 interactions, MDSC-TNF instigates (a) feed-forward Cxcl1 production in tumor cells; (b) inflammatory cancer-associated fibroblast (CAF) polarization, which further drives chemoresistant IL6-STAT3 signaling in PDAC tumor cells; and (c) T-cell dysfunction. This signaling circuitry sustains an immunosuppressive myelo-enriched and T-cell excluded microenvironment to dictate therapeutic resistance.