Figures & data
Figure 1. Ultrastructural and molecular mechanisms of bacterial endocytosis by intestinal epithelial cells. A brush border (BB), composed of densely packed actin-cored microvilli rooted in the terminal web (TW), serves as a barrier to prevent physical contact between luminal microbes and cellular soma. After low dose interferon gamma (IFNγ) stimulation, myosin light chain kinase (MLCK)-dependent phosphorylation of myosin light chain (MLC) in the TW region causes contraction and arc formation, leading to BB fanning. The consequently enlarged intermicrovillous cleft exposes the bacteria to cholesterol-rich lipid microdomains situated at the BB membrane invagination, and allows bacteria to be internalized via lipid raft- and caveolin-1-dependent endocytic pathways.
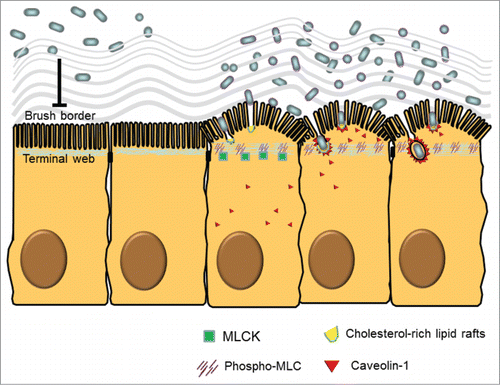
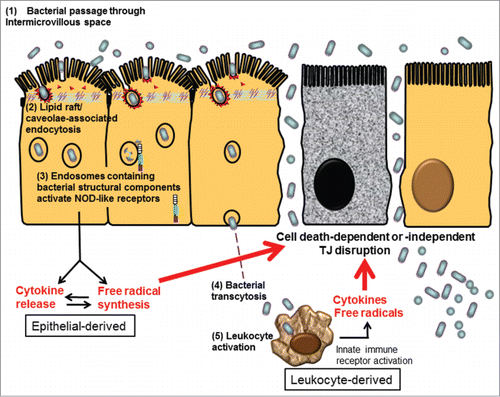
Figure 2. Proposed schema of bacterial endocytosis preceding tight junction damage in epithelial cells. (1) Bacteria passed through the widened intermicrovillous cleft and were exposed to lipid rafts on apical membrane invagination. (2) Bacterial endocytosis by epithelial cells was mediated by lipid raft/caveolae-dependent pathways. (3) Cytosolic innate immune receptors, such as nucleotide-binding oligomerization domain (NOD)-like receptors (NLRs), were recruited to the membrane of endosomes containing microbes and bacterial structural components. Activation of NLR-dependent signaling leads to epithelial synthesis of proinflammatory cytokines and free radicals, aiming for bacterial elimination. However, excessive inflammatory or oxidative stress may act as a double-edged sword causing cell death-dependent or -independent TJ disruption in epithelial cells. (4) Internalized bacteria, if managed to evade intracellular killing, may transcytose to the lamina propria. (5) Transcytosed bacteria activate leukocytes, resulting in even higher levels of proinflammatory cytokines and free radicals to aggravate TJ damage, leading to uncontrollable inflammation.