Figures & data
Figure 1. The variation of color changes in tested compounds (A) before and (B) after the process of reduction of Ag+ to Ag0 nanoparticles. (C) UV–Vis absorption spectra of AgNPs synthesized using cell-free supernatant of Bacillus sp. (A15 and D29) after 24 h.
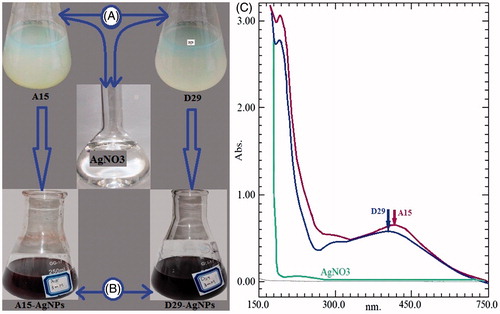
Figure 2. The FT-IR spectra of AgNPs biosynthesized by cell-free supernatant of Bacillus amyloliquefaciens (D29) and Bacillus subtilis (A15).
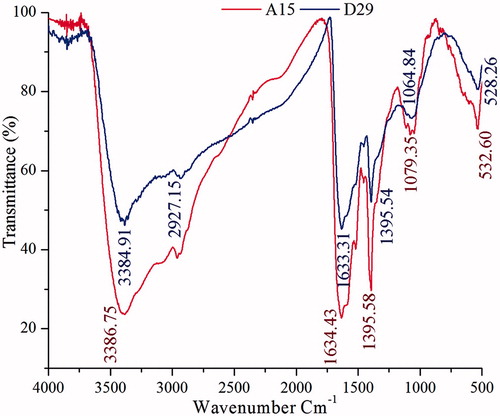
Figure 3. SEM images of AgNPs synthesized by reducing 3 mM AgNO3 using cell free supernatant of B. amyloliquefaciensD29 (A) and B. subtilisA15 (B); XRD patterns of synthesized AgNPs (C and D).
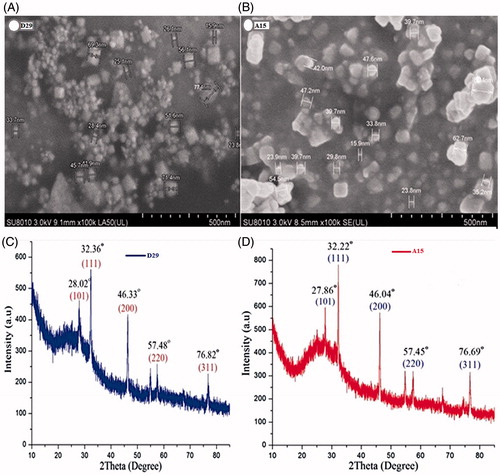
Figure 4. (A) TEM images of AgNPs synthesized by reducing 3 mM AgNO3 using cell free supernatant of B. subtilis (A15) and B. amyloliquefaciens (D29), (B) EDX patterns of synthesized AgNPs.
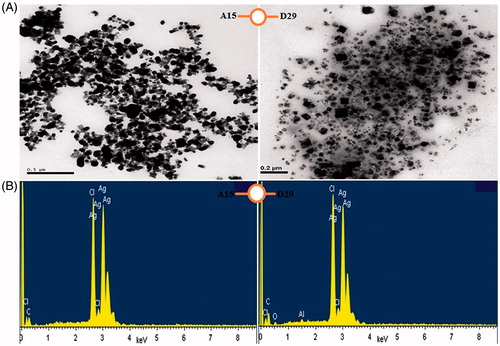
Figure 5. Graph showing the schematic representation of the biomediated synthesis of silver nanoparticle. Silver nitrate (AgNO3) ionises to silver ion (Ag+) and [(NO3)−1]. The bacterial cell wall has loosely extracellular polymeric substance (EPS).
![Figure 5. Graph showing the schematic representation of the biomediated synthesis of silver nanoparticle. Silver nitrate (AgNO3) ionises to silver ion (Ag+) and [(NO3)−1]. The bacterial cell wall has loosely extracellular polymeric substance (EPS).](/cms/asset/a82e2ba3-4b0f-4889-824a-3905c58d78ad/ianb_a_1241793_f0005_c.jpg)
Table 1. Larval and pupal toxicity of silver nanoparticles synthesized using cell free supernatant of B. amyloliquefaciens (D29) against C. pipiens pallens.
Table 2. Larval and pupal toxicity of silver nanoparticles synthesized using cell-free supernatant of B. subtilis (A15) against C. pipiens pallens.
Figure 6. (A) Mortality of Cx. pipiens pallens mosquito larvae (fourth instar) after 24 h exposure of AgNPs synthesized using cell free supernatant of B. subtilis and B. amyloliquefaciens. (B) No mortality was observed in control larvae. (C) Light micrographs showing morphological characteristics of treated larvae which represent morphological changes in the head, thorax, abdomen and anal papillae region. (D) No morphological changes were observed in the head, thorax and abdomen region of control larvae. (E) The treated pupae showed considerable morphological changes in the whole body spatially in the cuticle layer and trumpet. (F) The control pupae showed no morphological disruption.
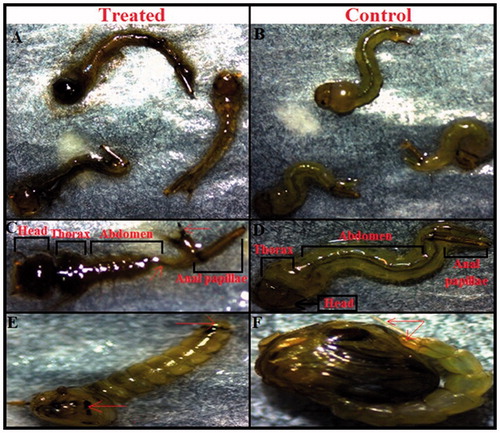
Figure 7. Mechanism of silver nanoparticle in Hemolymph (insect fluid) and cuticle (A) Chemical structure of Hemolymph and cuticle before treatment, (B) Silver nanoparticle (AgNPs) attached with Hemolymph and cuticle after treatment.
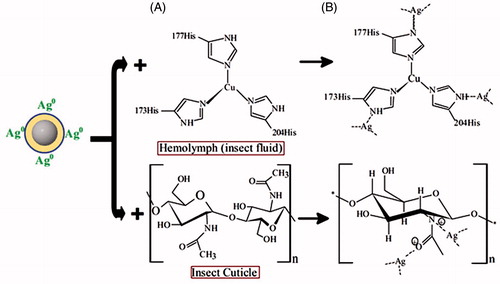
Figure 8. Zones of inhibition of reaction mixture containing silver nanoparticles and B. subtilis A15 and B. amyloliquefaciens D29 against Xanthomonas oryzae pv. oryzae growth on LBA medium.
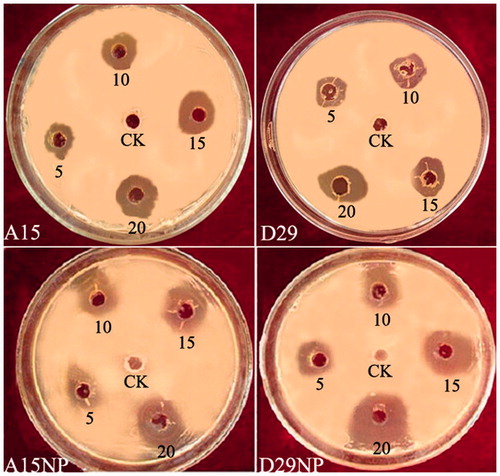
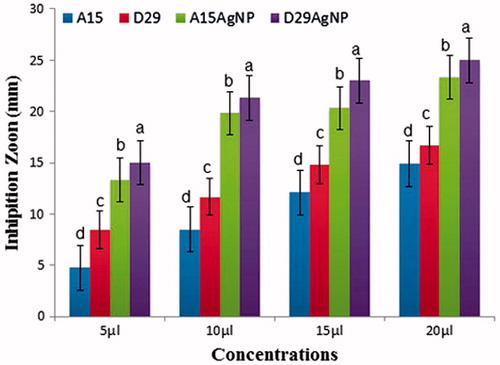
Figure 9. In vitro inhibitory effect (mm) of two bacterial strains (A15 and D29) and (AgNPs) against Xanthomonas oryzea growth on Luria-Bertani agar (LBA) medium after 48 h incubation at 28 ± 2 °C in laboratory. Columns with the different letters indicate significant differences (P < .05) among treatments, Error bars in each column represent the standard error within same treatment.