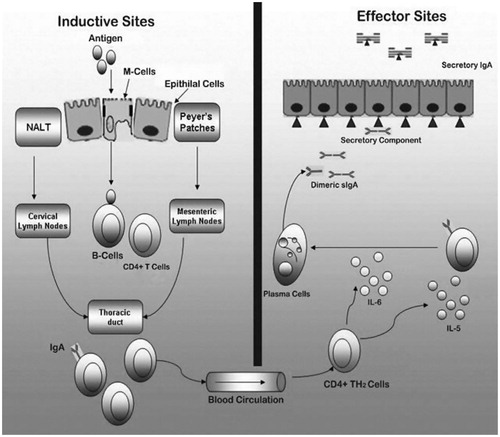
Figures & data
Table 1. Comparison of parenteral and mucosal administration of vaccine.
Table 2. Classification of mucoadhesive polymers.
Table 3. List of patent on biodegradable/mucoadhesive polymers used for vaccine delivery.
Table 4. List of patent on ligands decorated system for vaccine delivery.
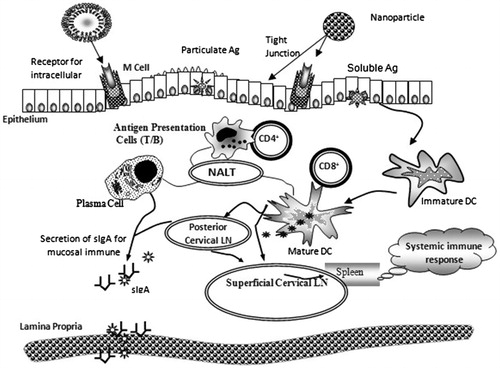
![Figure 3. Schematic representation of targeting potential of surface-modified over non-modified nanocarriers [Citation19].](/cms/asset/4ba9d412-71a9-4a45-8aeb-02dd8998fc98/ianb_a_1345927_f0003_b.jpg)