Figures & data
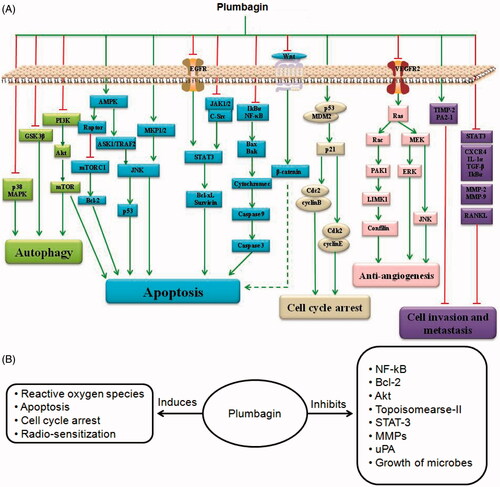
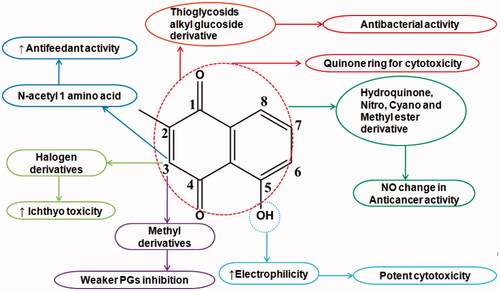
Table 1. Biological activities and mechanism of actions of plumbagin.
Table 2. The pharmacokinetic parameters of plumbagin after oral administration of 100 mg/kg body weight of plumbagin to healthy male Wistar rats [Citation3].
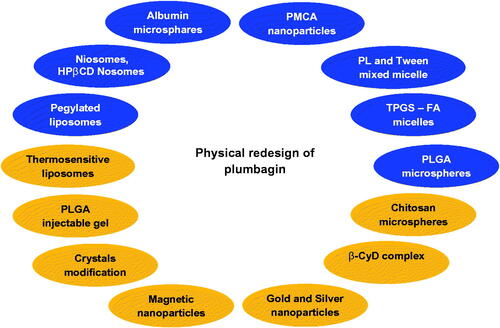
Table 3. Various novel drug delivery systems of plumbagin along with their key findings.