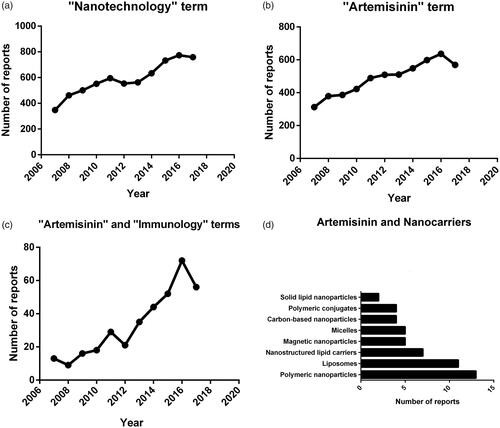
Figures & data
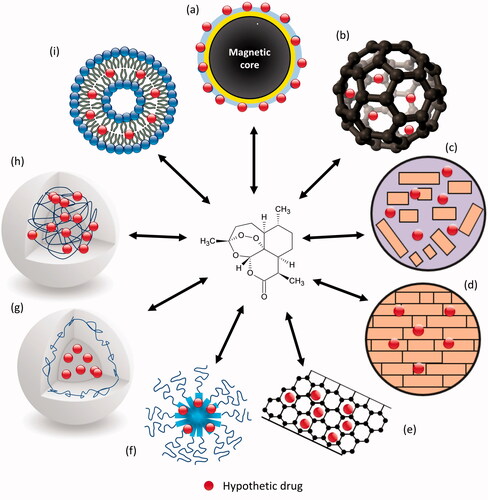
Table 1. Summary of the main works with polymeric nanocarriers and artemisinin and its derivatives.
Table 2. Summary of the main works with lipid nanocarriers and artemisinin and its derivatives.
Table 3. Summary of the main works with carbon-based and magnetic nanoparticles and artemisinin and its derivatives.

![Figure 1. Schematic representation of immunomodulatory suppressive effects of artemisinin in B and T cells. (a) The expansion of regulating T cells, increasing immune tolerance and activation of cytotoxic T cells and (b) Inhibition of Th1 and Th17 responses, decrease the release of matrix metalloproteinases (MMPs) and TNF-α; Inhibition of IgG production by B cells. Adapted from Shakir and co-workers [Citation18].](/cms/asset/dce14f2d-9b77-420e-9f2d-cc1cc4998053/ianb_a_1505739_f0001_c.jpg)