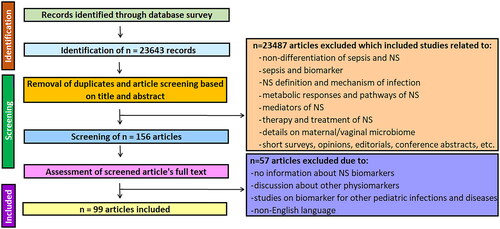
Figures & data
Table 1. Major differences in EOS and LOS.
Table 2. The diagnostic parameters of biomarkers for the identification of neonatal sepsis.
Table 3. Potential genes and miRNA based biomarkers for the diagnosis of NS.
Table 4. Recent Advances in developing electrochemical biosensor to diagnose neonatal sepsis.
![Figure 1. Schematic representation of fabricating electrochemical sensor to detect neonatal sepsis biomarker. [Abbreviations - CV: Cyclic voltammetry, EIS: electrochemical impedance spectroscopy, DPV: differential pulse voltammetry, SWA: square wave voltammetry, CA: chronoamperometry, POC: point of care].](/cms/asset/a044ee8d-758b-499e-bf09-b52edd292535/ianb_a_2252016_f0001_c.jpg)
![Figure 3. Schematic representation of different diagnostic biomarker expressions due to bacterial infection in neonatal sepsis. [Abbreviations - SAA: serum amyloid A, CRP: C reactive protein, LBP: Lipopolysaccharide Binding Protein, PCT: procalcitonin, IL: interleukin].](/cms/asset/90577550-54be-4897-8594-351e5e6724d2/ianb_a_2252016_f0003_c.jpg)