Figures & data
Figure 1 Patient disposition.
Abbreviations: BD, budesonide; BFF, budesonide/formoterol fumarate; FEV1, forced expiratory volume in 1 second; FF, formoterol fumarate; MDI, metered dose inhaler.
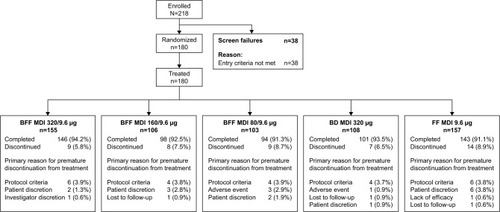
Table 1 Baseline demographics and clinical characteristics (safety/ITT population)
Figure 2 FEV1 AUC0–12 on Day 29 (mITT population).
Abbreviations: AUC0–12, area under the curve from 0 to 12 hours; BD, budesonide; BFF, budesonide/formoterol fumarate; FEV1, forced expiratory volume in 1 second; FF, formoterol fumarate; LSM, least squares mean; MDI, metered dose inhaler; mITT, modified intent-to-treat.
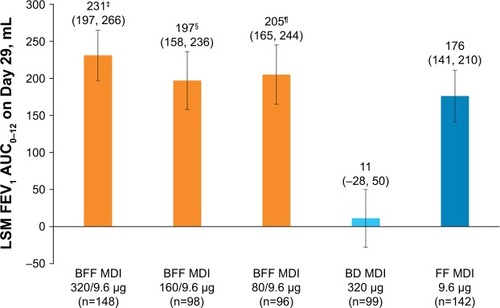
Table 2 Summary of secondary efficacy endpoints (mITT population)
Figure 3 Peak change from baseline in FEV1 over 28 days (mITT population).
Abbreviations: BD, budesonide; BFF, budesonide/formoterol fumarate; FEV1, forced expiratory volume in 1 second; FF, formoterol fumarate; LSM, least squares mean; mITT, modified intent-to-treat; MDI, metered dose inhaler.
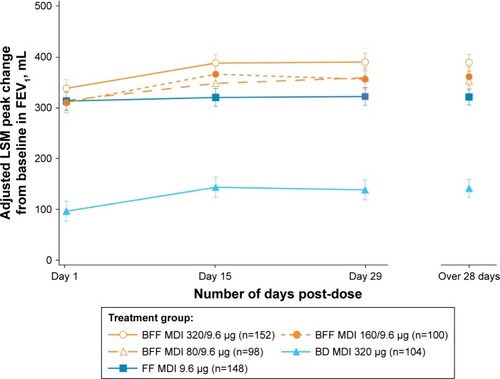
Table 3 Summary of TEAEs overall and by treatment group (safety population)Table Footnotea
Figure 4 Relative bioavailability of budesonide and formoterol on Day 29 (PK population).
Abbreviations: AUC0–12, area under the curve from 0 to 12 hours; BD, budesonide; BFF, budesonide/formoterol fumarate; Cmax, maximum observed plasma concentration; FF, formoterol fumarate; LSM, least squares mean; MDI, metered dose inhaler; PK, pharmacokinetic.

