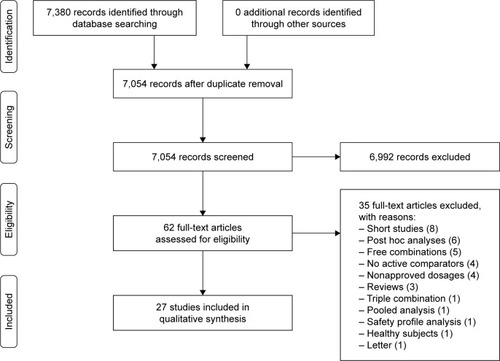
Figures & data
Table 1 Study characteristics and investigated outcomes
Table 2 Synthesis of the main pharmacological characteristics of LABAs, LAMAs, and ICS investigated in the clinical trials included in this systematic review
Table 3 Studies in which LABA/LAMA combinations were significantly superior ↑, similar ≈, or inferior ↓ to comparators