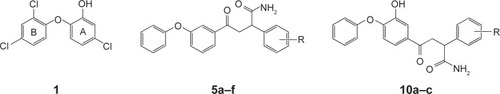
Figures & data
Figure 2 The general synthetic route for the synthesis of compounds 5a–f.
Abbreviation: DMSO, dimethyl sulfoxide.
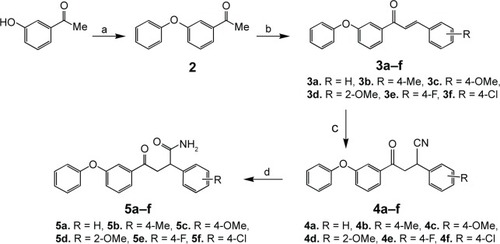
Figure 3 The general synthetic route for the synthesis of compounds 10a–c.
Abbreviation: DMSO, dimethyl sulfoxide.
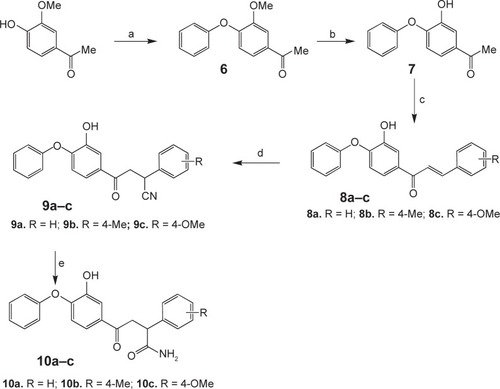
Table 1 In vitro antitubercular activity (MIC), cytotoxicity (CC50), SI, Clog P, and PSA of compounds 5a–f and 10a–c
Table 2 Molecular docking results of compounds 5a–f and 10a–c
Figure 4 Molecular docking interaction of compound 10b (green) with Mtb ENR (PDB 1P45).
Abbreviations: Mtb ENR, enoyl-acyl carrier protein reductase of Mycobacterium tuberculosis; NAD, nicotinamide adenine dinucleotide.
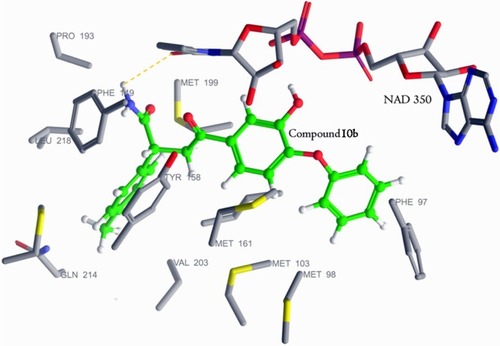
Table 3 Evaluation of druglikeness of compound 10b
Table 4 Human microsome stability study of compound 10b