Figures & data
Table 1 Formulation of ITO HCl floating tablets
Table 2 Properties of the prepared tablet
Table 3 Floating properties of ITO HCl floating tablets
Figure 2 Dissolution profiles of ITO HCl floating tablets F1 to F10.
Abbreviations: ITO, itopride; HCl, hydrochloride; SD, standard deviation; HPMC, hydroxypropyl methylcellulose; EC, ethylcellulose; Eud, eudragit.
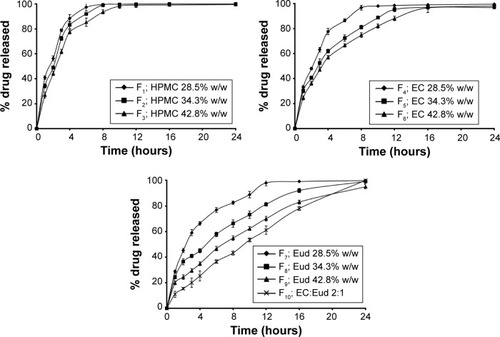
Table 4 Kinetic treatment of the dissolution data for ITO HCl floating tablets
Figure 3 Plasma concentrations of ITO HCl after oral administration of the commercial tablets and the prepared ITO HCl sustained-release tablet formula F10.
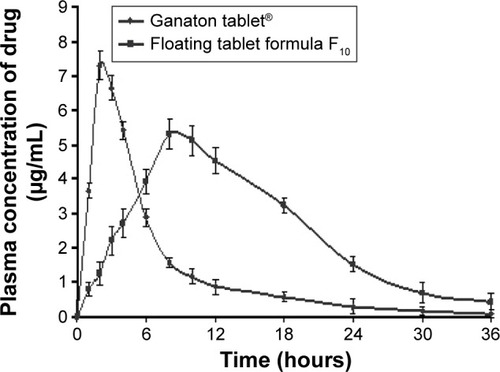
Table 5 Pharmacokinetic parameters of ITO HCl following oral administration of the prepared ITO HCl sustained-release tablets formula F10 in comparison with the commercial immediate-release tablets, the commercial tablets
Table 6 Physical properties of the aged batches of floating tablets after shelf storage for 6 months
Figure 4 Effect of aging on the release of ITO HCl from floating tablet formula F10.
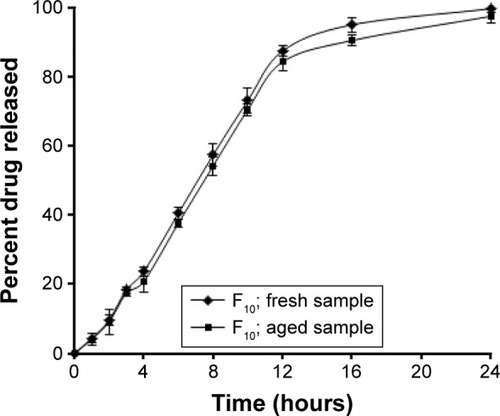
Table 7 ITO HCl content in the selected floating tablets stored for 6 months at different temperatures (30°C, 40°C, and 50°C) and at 75% RH
Table 8 First-order degradation kinetics parameters of ITO HCl in different formulae after storage for 6 months at different temperatures

