Figures & data
Table 1 Demographic characteristics of the study population
Table 2 Summary of pharmacokinetic parameters for CKD-519 after administration of single oral dose in healthy volunteers
Figure 2 Mean plasma CKD-519 concentration–time profiles after administration of ascending single oral doses in healthy male subjects.
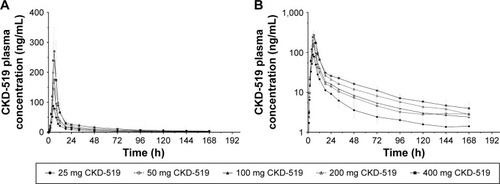
Figure 3 Mean (± standard deviation) percent change of CETP activity from baseline–time profiles after administration of ascending single oral doses in healthy male subjects.
Abbreviation: CETP, cholesteryl ester transfer protein.
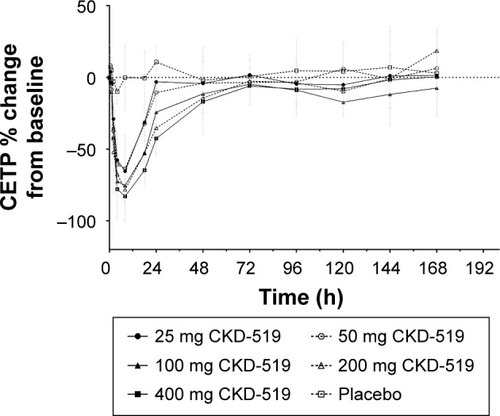
Figure 4 Relationship between CETP activity and CKD-519 concentration after administration of single oral doses of CKD-519 (from 25 mg to 400 mg) in healthy male subjects.
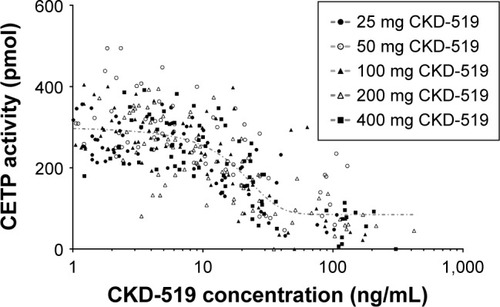
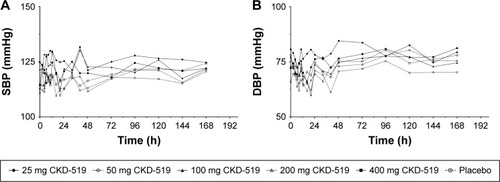
Figure 5 Blood pressure changes after administration of single oral doses of CKD-519 (from 25 to 400 mg) in healthy male subjects.
Abbreviations: SBP, systolic blood pressure; DBP, diastolic blood pressure.