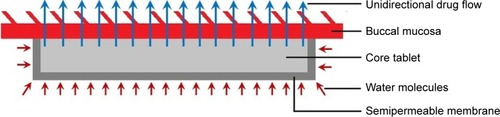
Figures & data
Table 1 Composition of BH.2HCl mucoadhesive buccal tablets
Table 2 Physicochemical parameters of the developed BH.2HCl mucoadhesive buccal tablets
Figure 2 Plot of % swelling vs time for BH.2HCl mucoadhesive buccal formulations (A) F1–F6 and (B) F7–F12.
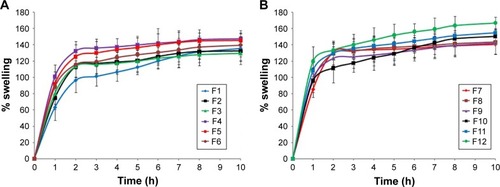
Table 3 Mucoadhesive strength, force and residence time of BH.2HCl mucoadhesive buccal tablets
Table 4 In vivo mucoadhesion properties of BH.2HCl mucoadhesive buccal tablets
Figure 3 Release profile of BH.2HCl from different mucoadhesive buccal formulations: (A) F1–F6 and (B) F7–F12.
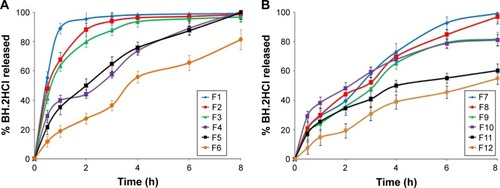
Table 5 Ex vivo permeation parameters of BH.2HCl mucoadhesive buccal tablets
Figure 4 Permeation profile of BH.2HCl from different mucoadhesive buccal formulations.
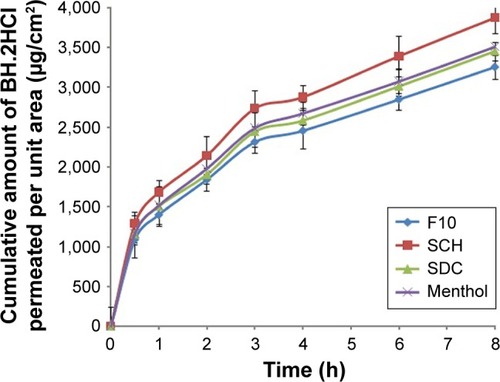
Figure 5 DSC thermograms of (A) pure BH.2HCl, (B) guar gum, (C) Na CMC, (D) HPMC K4M, (E) mannitol, (F) Aerosil 200, (G) magnesium stearate (H) SCH and (I) formulation F10 in addition to SCH.
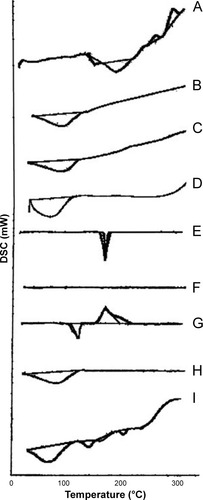
Figure 6 FTIR spectra of (A) pure BH.2HCl, (B) guar gum, (C) Na CMC, (D) HPMC K4M, (E) mannitol, (F) Aerosil 200, (G) magnesium stearate (H) SCH and (I) formulation F10 in addition to SCH.
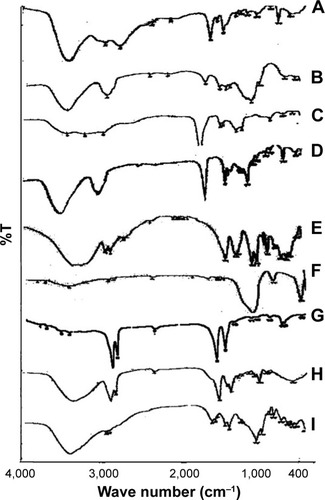
Figure 7 Chromatogram of BH.2HCl in the mobile phase at λmax 260 nm.
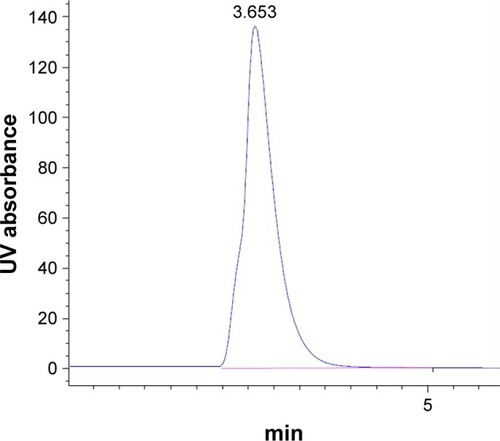
Table 6 Percentage of BH.2HCl remained in formulation F10 when stored at different temperatures (40°C and 60°C) for 12 weeks
Figure 8 Plot of log % BH.2HCl remained vs time after storage of the optimized buccal formulation for 12 weeks at 40°C and 60°C according to first-order kinetics.
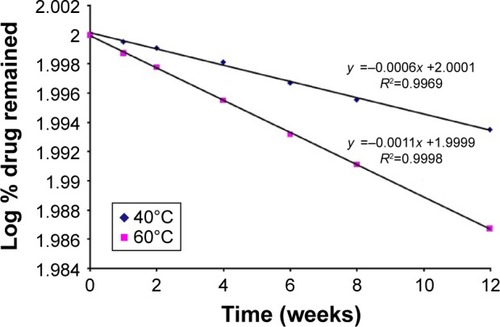
Figure 9 BH.2HCl plasma concentrations following the administration of the optimized buccal formulation and oral tablets Betaserc® 24 mg.
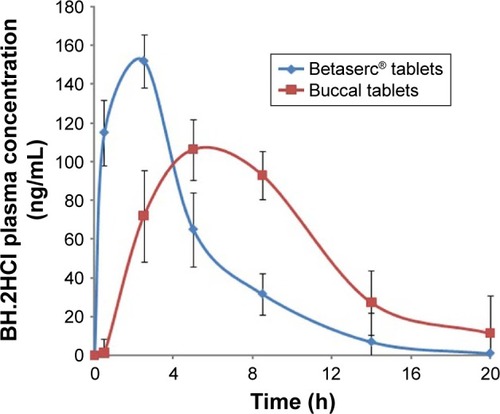
Table 7 Mean pharmacokinetic parameters for the volunteers following administration of the optimized mucoadhesive buccal formulation and oral Betaserc® tablets