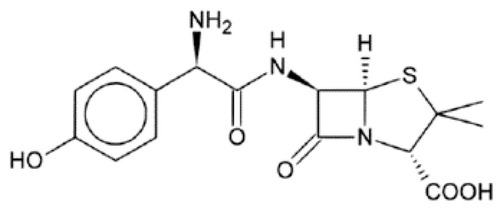
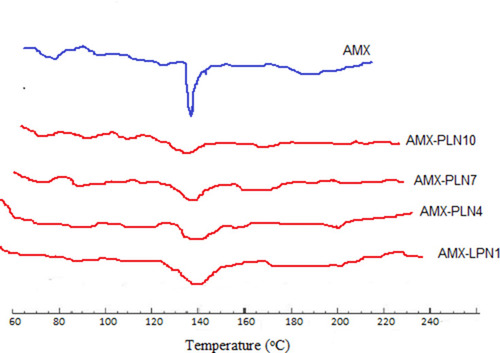
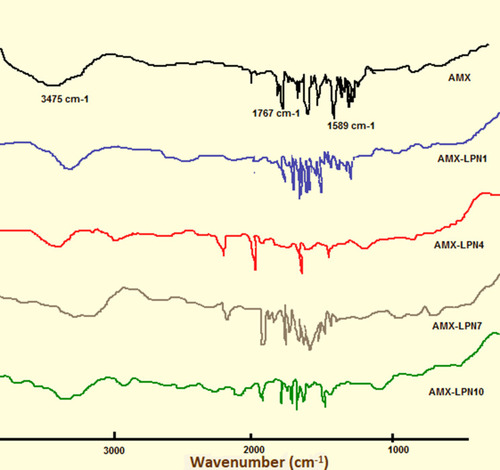
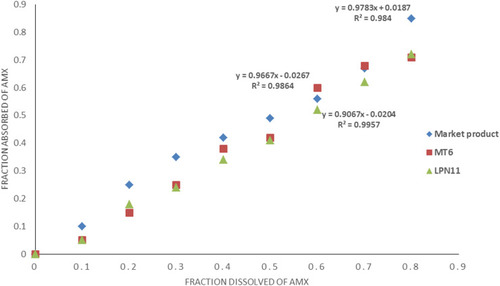
Figures & data
Table 1 Composition and Physical Character of AMX-MTs Formulation, Each Dose of Tablets Contain 250 Mg of AMX
Table 2 Formulation Code, Drug:Lipid Ratio and Chitosan (CN) Solution Concentration (mg/mL), PI, ZP %EE and % DL of AMX-LPN Formulations
Figure 5 (A, B) In vitro dissolution profiles of AMX from Mini-tablets formulations (A) for MT1-MT-6, (B) for MT-7-MT-12 in 0.1N HCl. Each point is a mean value of three tablets.
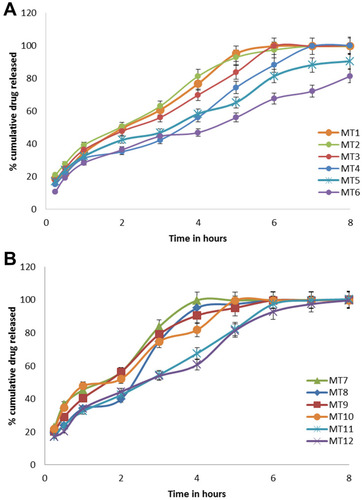
Figure 6 (A-D). In vitro dissolution profiles of AMX from LPNs formulations for (A) LPN1-3, (B) LPN4-6, (C) LPN7-9, (D) LPN10-12 in 0.1N HCl. Each point is a mean value of three trials.
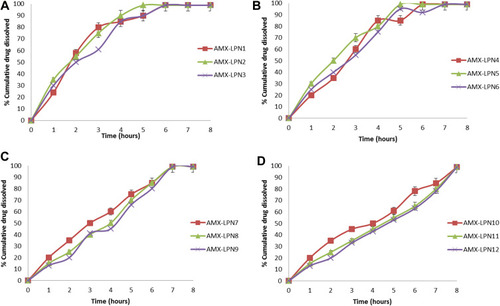
Table 3 Study of in vitro Release Kinetics Data for AMX from Selected Mini-Tablets and Lipid/Polymer Nanolipid Systems
Table 4 %EE, PS, ZP of AMX-LPN11 Under Different Storage Conditions for 45 Days
Table 5 Mean Results ±SD for Microbiological Tests of Selected AMX-PLN Formula
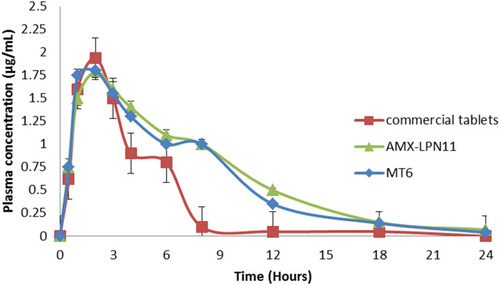
Table 6 Pharmacokinetic Parameters of AMX After Oral Administration of AMX-LPN11 and Commercial Tablets in Rats