Figures & data
Figure 1 Structure of somatostatin 14 (A) and somatostatin 28 (B). The 2 isoforms differ in the amino acids within the red dashed rectangle, as they are missing in somatostatin 14 but are present in somatostatin 28.
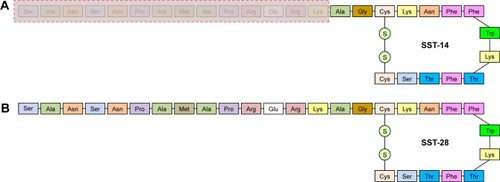
Figure 2 Structure of the most used somatostatin analogs: octreotide (A), lanreotide (B), vapreotide (C), and pasireotide (D). Lanreotide is an octapeptide that self-assembles into nanotubes when placed in an aqueous environment. It has 3 aromatic residues (d-naphthylalanine, tyrosine, and d-tryptophan) that are involved in the formation of these supramolecular structures.
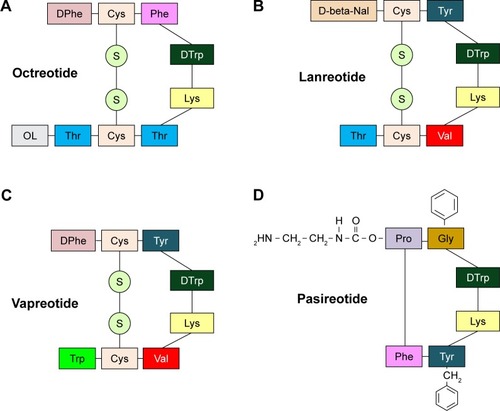
Figure 3 Mechanisms explaining the inhibitory and antiproliferative effects of SST and SSAs. There are 5 types of SSTRs, which show different tissue distribution. SSTR 2 has 2 different isoforms (2a and 2b) that derive from alternative splicing at the C-terminus of the receptor – between the 2, the SSTR 2a isoform is far more common in human tissues. SST and SSAs bind to SSTRs with different affinities. All SSTRs are G protein-coupled receptors with 7 transmembrane-spanning domains.
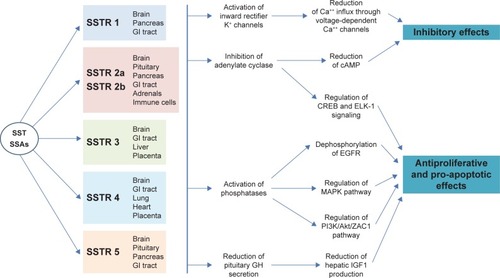
Table 1 Classifications of GEP-NENs
Table 2 Relevant clinical studies of LAN Autogel® in GEP-NENs
