Figures & data
Figure 2 The introduction of functional groupings in various MSN regions: (a) at the external surface, (b) at the pore entrances, or (c) within the walls. Data from Slowing II et al.Citation56
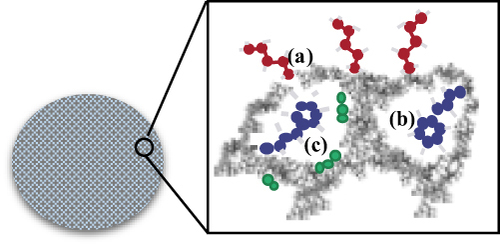
Figure 3 The endotemplate approach for porous materials (soft matter templating). Data from Niesz et al.Citation59
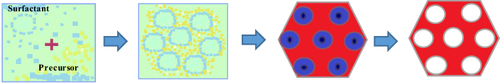
Figure 4 Schematic illustration for the production of MSNs grafted with a copolymer of positively charged quaternary amines and PEG using RAF and addition-fragmentation equilibria during RAFT polymerization. (I) Immobilization of the RAFT agent using the equal molar quantity of diisopropylcarbodiimide(DIC) and diisopropylethylamine(DIPEA) as catalysts, (II) The RAFT polymerization of PEGMA and PDMAEA at 70 °C under an argon atmosphere in the presence of 2.2′-azobis(isobutyronitrile) (AIBN), and (III) The final product, namely as MSN-PEG+. Data from Sahoo et al.Citation65
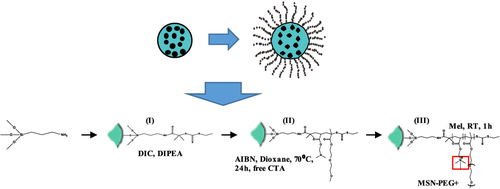
Figure 5 An example of how a dual-responsive system behaves in watery medium. Data from Paris et al.Citation69
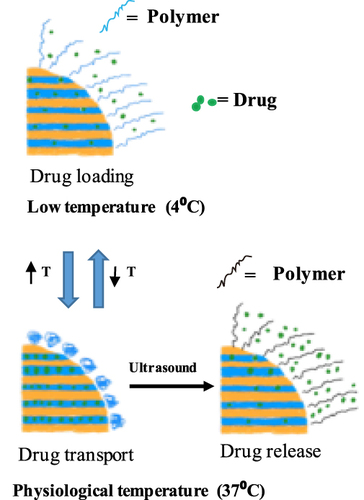
Figure 6 Synthesis of a dual-responsive drug delivery system illustrated. Data from Zheng et al.Citation70
Table 1 Poorly Water-Soluble Anticancer Drugs Encapsulated into Mesoporous Silica
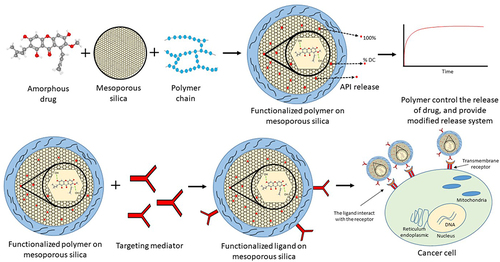
Figure 7 The mechanism of drug release improvement from the drug within non-functionalized mesoporous silica.
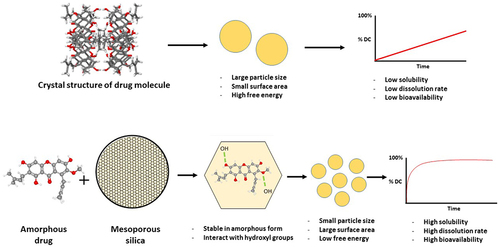
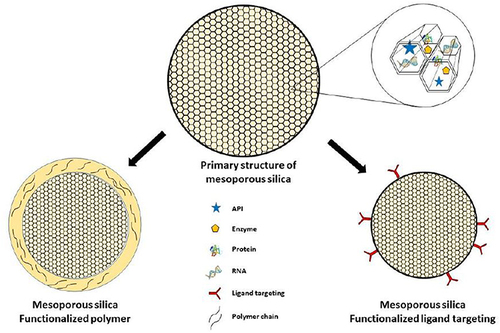
Figure 8 Functionalized polymer/ligand on mesoporous silica.