Figures & data
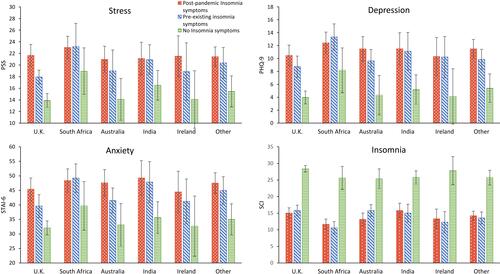
Figure 1 The schematic illustration of the basic neuronal organization of the paraventricular nucleus (PVN) of the hypothalamus depicting its purported role in a four-step model of neurocircuitry of anxiety. On the left, a four-step model by Calhoon and Tye is shown: according to this model external events are detected, interpreted, evaluated, and responded to by succeeding levels of highly interconnected neural circuits. Reproduced by permission from Springer Nature, Nat Neurosci, Resolving the neural circuits of anxiety, Calhoon GG, Tye KM. 2015;18(10):1394–1404. Copyright © 2015, Nature Publishing Group, a division of Macmillan Publishers Limited.Citation92 Events are further interpreted as threatening or nonthreatening depending on the balance between opposing circuits; when the balanced is shifted toward projections interpreting events as threatening, this leads to anxiety. On the right, PVN’s neurocircuitry is depicted. Afferent inputs to the nucleus arrive from many important integrative centers of the medulla, pons and hypothalamus. Subfornical organ is among major inputs with identified angiotensin release acting directly on magnocellular and parvocellular neurons, or indirectly via intranuclear circuitry that includes inhibitory Gamma-aminobutyric-acid (GABA) (red) and excitatory glutamate (green) interneurons. Changes within paraventricular angiotensin subcircuitries are described under stress condition and are reflected on release of stress hormone and arousal. GABA interneurons have been shown to express angiotensin-converting-enzyme-2 (ACE2),Citation93 and as such might be targeted by severe-acute-respiratory-syndrome-coronavirus-2 (SARS-CoV-2). The majority of GABA interneurons have been localized to the halo zone surrounding the PVN, and their role as an additional gatekeeper and integrator in controlling the excitability of PVN outputs has been proposed.Citation94 Any changes in the PVN circuitries, due to their major control over most of neuro-endocrine axes and neuronal autonomic centers may cause robust alteration in homeostatic regulation, and through influence on regulatory brain centers impact on sleep and wakefulness, increased propensity to trigeminal autonomic cephalalgias and alteration of ultradian rhythms.
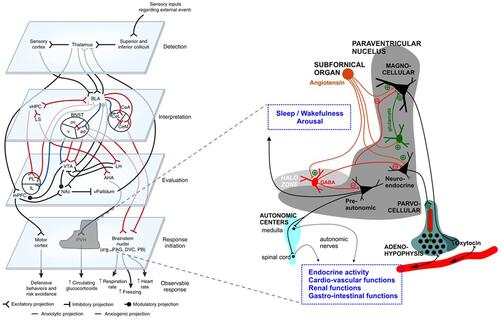
Figure 2 Stress, depression, anxiety, and insomnia symptoms across countries with the top five number of respondents by insomnia group (n = 2525). Error bars indicate 95% confidence intervals.