Figures & data
Table 1 UICC TNM Staging of Kidney Tumours
Table 2 Leibovich Risk Score
Table 3 UCLA Integrated Staging System (UISS)
Table 4 Summary of Early Clinical Studies Investigating Adjuvant RCC Therapies
Figure 1 Mechanism of action of TKIs used in metastatic RCC. Kinases regulating angiogenesis are frequently overexpressed in RCC, culminating in increased tumour vascular supply. VHL inactivation leads to overexpression of pro-angiogenic factors including VEGF via increased HIFα expression. HIFα is also upregulated via PI3K/mTOR signalling. VEGF binds to VEGFR1/2/3 on endothelial cell surfaces, promoting angiogenesis. Additional cell surface receptors regulating angiogenesis include PDGFR (platelet-derived growth factor receptor), FGFR (fibroblast growth factor receptor), tyrosine-protein kinase MET (hepatocyte growth factor receptor), AXL oncogene (tyrosine-protein kinase receptor UFO) and proto-oncogene tyrosine-protein kinase receptor RET. A range of TKIs have been developed targeting various aspects of these signalling pathways. Adapted by permission from Springer Nature © (2017). Posadas EM, Limvorasak S, Figlin RA. Targeted therapies for renal cell carcinoma. Nat Rev Nephrol. 2017;13(8):496–511.Citation13
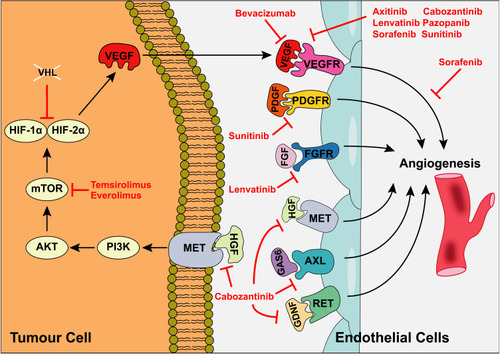
Table 5 Summary of Newer Generation Studies Testing Adjuvant TKIs
Figure 2 Mechanism of action of ICIs. In response to recognition of TA presented by cancer cells, T-cell activation depends on coalescence of a range of signals including co-stimulation by CD80/CD86 which bind to CD28 on the T-cell. Cancer cells evade the immune response via competitive binding of CD80/CD86 to CTLA-4 and via binding of PD-L1/PD-L2 to PD-1, inhibiting T-cell activation. Antibodies targeting CTLA-4, PD-1 and PD-L1 prevent activation of immunosuppressive signals, thus enhancing host immune responses against cancer cells. Adapted by permission from Springer Nature © (2020). Xu W, Atkins MB, McDermott DF. Checkpoint inhibitor immunotherapyin kidney cancer. Nat Rev Urol. 2020;17(3):137–150.Citation16
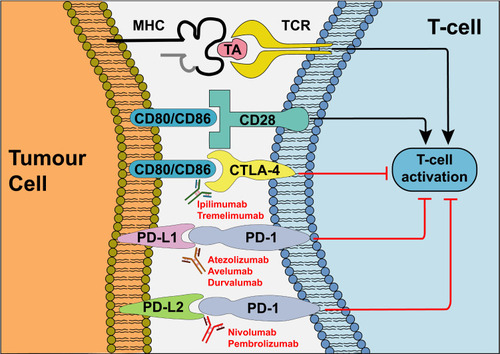
Table 6 Summary of Newer Generation Studies Testing Adjuvant ICIs
