Figures & data
Figure 1 Eslicarbazepine is formed by reduction of the acetate salt (shown on the right) or may be found as the predominant (4:1) active metabolite of oxcarbazepine (middle). Carbamazepine and its active epoxide metabolite are shown on the left. Reprinted with permission from Almeida L, Soares-da-Silva P. Eslicarbazepine acetate (BIA 2-093). Neurotherapeutics. 2007;4:88–96.Citation12 Copyright © 2007 Elsevier.
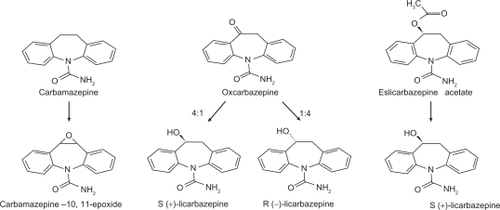
Table 1 Summary of potential pharmacokinetic interactions between eslicarbazepine and other medicationsCitation29
Table 2 Summary of percentage of adverse events across two placebo-controlled trialsCitation27,Citation33
Table 3 Eslicarbazepine levels as a function of doseCitation27