Figures & data
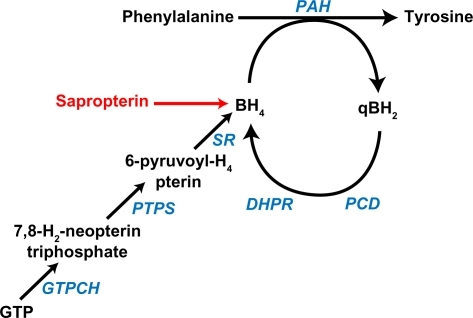
Figure 1 Phenylalanine hydroxylation. Phenylalanine is hydroxylated to tyrosine through the catalytic activity of phenylalanine hydroxylase, which requires the presence of the unconjugated pterin cofactor, tetrahydrobiopterin (BH4). Sapropterin is a synthetic form of BH4 that augments the endogenous BH4 supply. During phenylalanine hydroxylation, BH4 is oxidized to quinonoid dihydrobiopterin (qBH2). Fully active BH4 is regenerated through the sequential action of pterin-4a-carbinolamine dehydratase and dihydropteridine reductase (DHPR) or may be synthesized de novo from guanosine triphosphate (GTP).Citation2