Figures & data
Table 1. Composition of FDP-PIO bioadhesive buccal tablets.
Figure 1. Ex vivo permeation of (a) FDP and (b) PIO solution through porcine buccal mucosa (mean ± SD, n = 3).
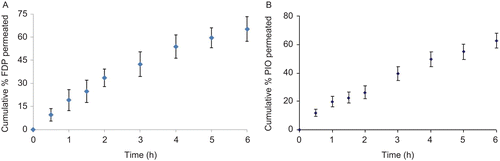
Table 2. Physicomechanical and bioadhesive properties of buccal tablets.
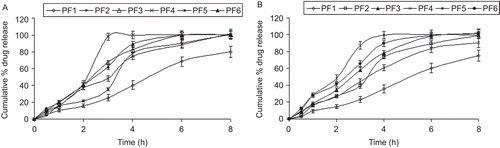
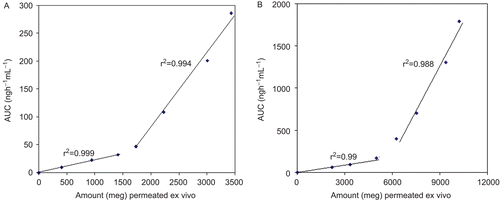
Table 3. FDP and PIO estimated values of release exponent (n) and correlation coefficients (r2), in combined dosage form for all of the formulations.
Figure 3. Mean serum profiles of (a) FDP and (b) PIO in pigs, after administration of oral solution and buccal tablet, values represented are mean ± SD (n = 6).
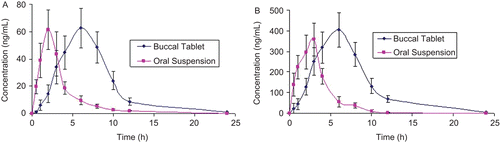
Table 4. Pharmacokinetic parameters of FDP and PIO in pigs after administration of oral suspension and bioadhesive buccal tablet, values represented are mean ± SD (n = 6).
Table 5. Stability study of the optimized formulation (PF6) for 3 months.