Figures & data
Table 1. Composition and evaluation of rosuvastatin sponge formulae prepared adapting a 41.31 full factorial design.
Table 2. Release kinetics and mechanism of the release data of rosuvastatin from the prepared sponges analyzed using DDsolver software.
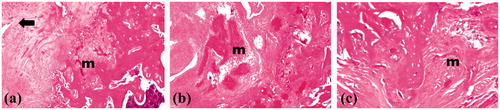
Figure 1. Photographic presentation of the surgical procedure (1) making an incision on the proximal anterior part of the femur, (2) Removing a 2-mm segment of the fractured femur and (3) fixing femur using an intramedullary K-wire.
Figure 2. Photographs of (a) xanthan gum-based, (b) polycarbophil-based, (c) Carbopol®-based and (d) Na alginate-based sponges at magnifications of 4×, all at 1:1 chitosan to anionic polymer ratio.
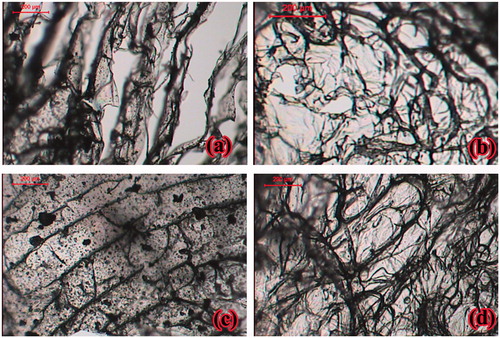
Figure 3. DSC thermograms of rosuvastatin, chitosan, anionic polymer, chitosan/anionic polymer physical mixtures and drug laoded sponges for (a) xanthan gum-based, (b) polycarbophil-based, (c) Carbopol®-based and (d) Na alginate-based formulae.
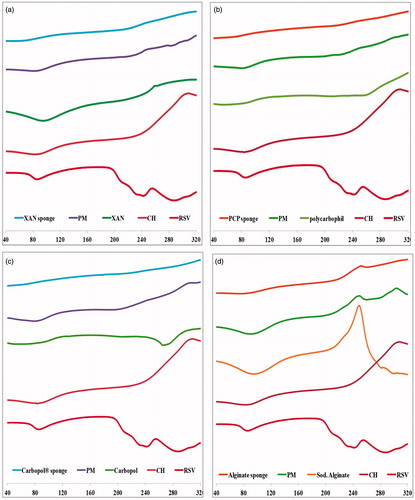
Figure 4. FT-IR of rosuvastatin, chitosan, anionic polymer, chitosan/anionic polymer physical mixtures and drug laoded sponges for (a) xanthan gum-based, (b) polycarbophil-based, (c) Carbopol®-based and (d) Na alginate-based formulae.
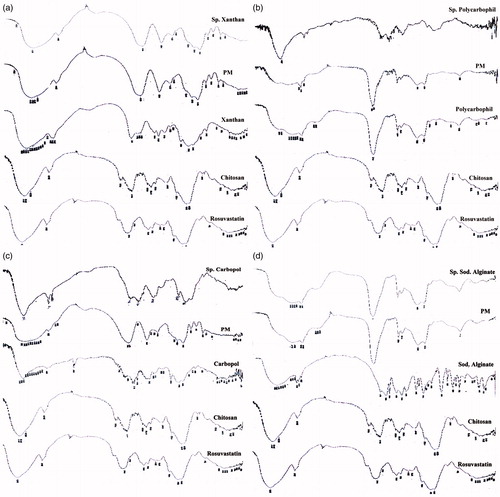
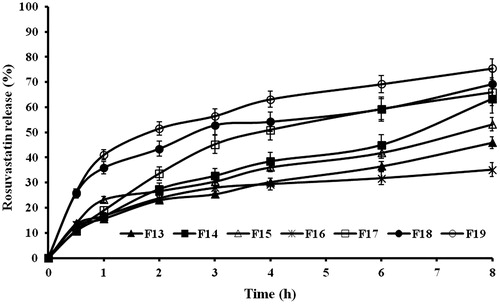
Figure 5. In vitro release profiles of rusovastatin from (a) xanthan gum-based, (b) polycarbophil-based, (c) Carbopol®-based and (d) Na alginate-based sponges prepared adapting a 41.31 full factorial design (n = 3, Error bars indicate SD).
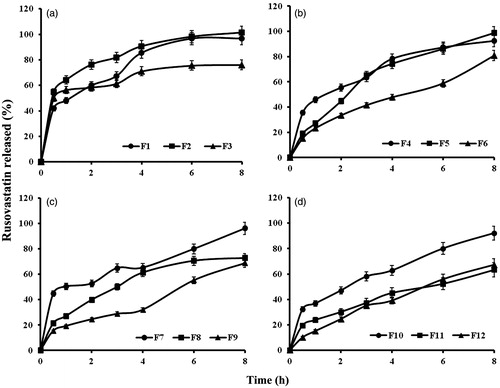
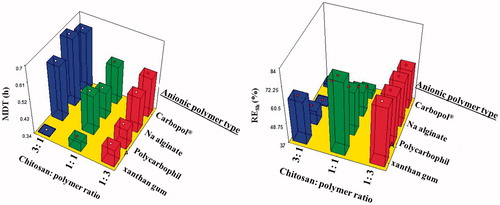
Figure 6. Effect of anionic polymer type and chitosan:polymer ratio on median dissolution time (MDT) and release efficiency (RE8h).