Figures & data
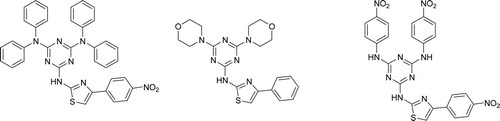
Figure 2. Hybrid diethylamino-chloro (series C) and diethylamino-dichloro (series D) phenylthiazolyl-s-triazine derivatives.
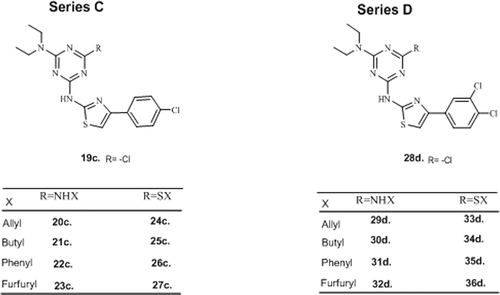
Table 1. Some physicochemical properties and spectral data of diethylamino-chlorophenylthiazolyl-s-triazine derivatives (series C)
Table 2 . Some physicochemical properties and spectral data of diethyl amino dichlorophenylthiazolyl-s-triazine derivatives (series D).
Scheme 1. Reagents and condition: (a) Na2CO3 0 -5°C; (b) 4-chloro-phenylthiazole-2-amine and 3,4-dichlorophenylthiazolyl-2-amine, NaHCO3, aqueous dioxane 45–50°C; (c) Alkenyl/alkyl/aryl/hetro alkyl–aryl amines or thiols in NaHCO3, dioxane 90–100°C under nitrogen atmosphere.
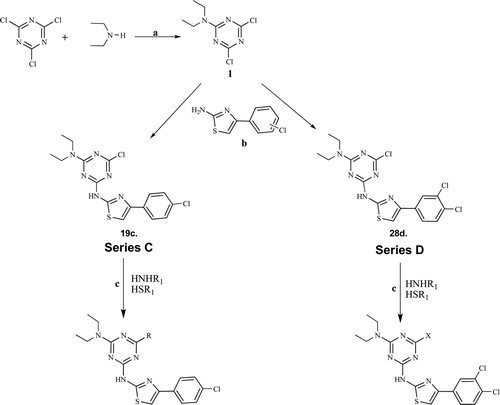
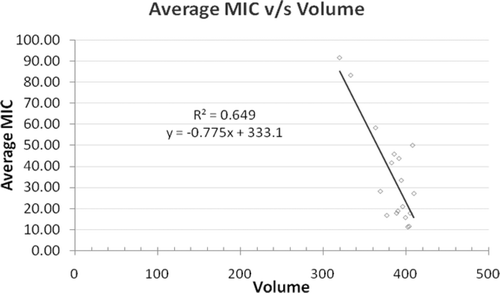
Table 3 . MIC values of series C and D against gram-positive/negative bacteria.
Table 4 . Brine shrimp cytotoxicity assay for compounds 31d and 32d.
Table 5 . Molinspiration calculations of compounds.
Table 6 . Osiris calculations of compounds.