Figures & data
Table 1. Inhibitory effects of 12a–f on the growth of human tumour cell lines expressed as IC50 in μg/mL.
Scheme 2. Synthesis 1,3,5-triazine derivatives bearing two 2-chloroethylamino residues 12e and three 2-chloroethylamino residues 12f.
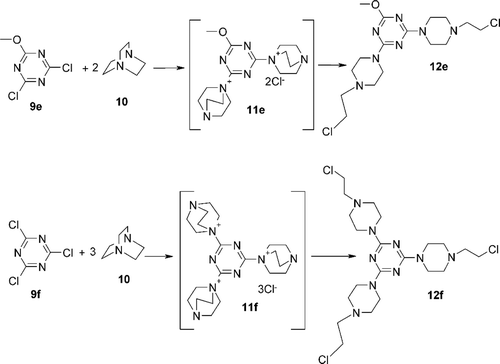
Table 2. Viability of MCF-7 cells treated for 24 h with different concentrations of chlorambucil (CHL) and compounds 12a–f.
Table 3. Alkylating activity of compounds 12a–f on comparison to chlorambucil; NBP test results.
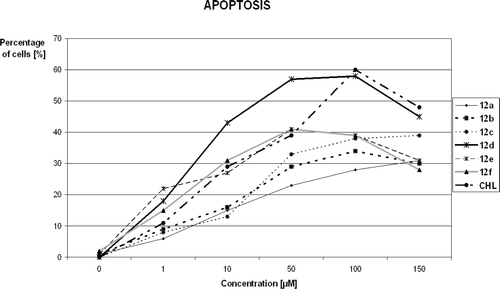
Figure 2. Percentage of apoptotic and necrotic MCF-7 cells after treatment with 50 μM solutions of chlorambucil (CHL) and compounds 12a–12f. 100% = {apoptotic(%) + necrotic(%) + viable cells}.
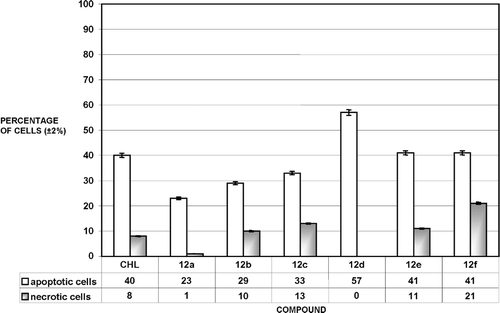
Table 4. DNA binding effect of compounds 12a–f.